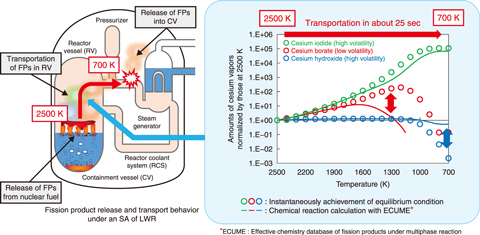
Fig.4-13 Amounts of cesium vapors transported to the lower temperature regions in a reactor under an SA calculated considering chemical reaction kinetics
Fission products (FPs) are released from nuclear fuel and transported into the containment vessel and environment through the high-temperature regions of the reactor vessel (hereafter, FP release and transport behavior) under a severe accident (SA) in a light water reactor (LWR). To enhance LWR safety, public exposure should be evaluated more accurately through a detailed estimation of FP release and transport behavior under an SA. The FP release and transport behavior is significantly influenced by FP chemistry, namely the kinds of reactions that take place and the chemical forms of the FPs. However, some of the existing SA analysis codes assume that equilibrium is instantaneously achieved, meaning that chemical reactions are assumed to process at infinite rates. Thus, there is large uncertainties on FP chemistry in the lower temperature regions, where the chemical reaction kinetics becomes slower.
A database of chemical reaction rate constants was therefore developed to estimate the FP chemistry during an SA considering chemical reaction kinetics. The developed FP chemistry database, ECUME, includes elemental models and thermodynamic data in addition to the chemical reaction rate constants for more accurate estimation of the FP chemistry. The chemical system of cesium and iodine was treated in ECUME due to their importance on public exposure to FPs under an SA. ECUME was the first documented database to include the data on boiling water reactor (BWR) control rod material boron which significantly affects the chemistry of cesium and iodine.
The amounts of cesium vapors during transportation from 2500 K to 700 K in a reactor under an SA were estimated using ECUME (Fig.4-13). The results were compared with those estimated using chemical equilibrium calculation applied to the existing SA analysis codes. Some vapors like cesium borate vapor showed large discrepancies between the data obtained by ECUME and those obtained by chemical equilibrium calculation. This indicates the possible over-estimation of low-volatile cesium borate deposits using the existing SA analysis codes, which can lead to the under-estimation of the amount of cesium released into the environment. Chemical reaction kinetics should thus be applied for more accurate evaluation of public exposure to FPs under an SA.
Future work will consist of experimentally validating ECUME, and expanding the ECUME to include other FPs like ruthenium to enhance the safety of reprocessing plants.