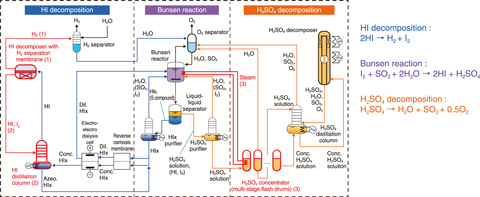
Fig.6-10 Flow diagram of the iodine-sulfur (IS) process
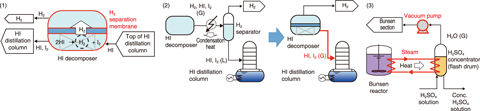
Fig.6-11 Innovative technologies for the IS process
The iodine-sulfur (IS) thermochemical hydrogen (H2) production process was investigated as to utilize heat from the High Temperature Gas-cooled Reactors. The IS process, depicted in Fig.6-10, consists of the Bunsen reaction to produce hydrogen iodide (HI) and sulfuric acid (H2SO4) (I2 + SO2 + 2H2O → H2SO4 + 2HI), HI decomposition to produce H2 (2HI → H2 + I2), and H2SO4 decomposition to produce oxygen (O2) (H2SO4→H2O + SO2 + 0.5O2). Overall, water is decomposed to produce H2 and O2.
Practical, competitive implementation of H2 production using the IS process requires improving the efficiency of the process for cost reduction. However, excess material flow is required due to the low reaction ratio, and the exothermic Bunsen reaction and material condensation generate large amounts of excess heat. As such, a high efficiency is difficult to achieve.
As such, technologies to improve the IS process efficiency have been investigated. A HI decomposition ratio higher than reaction equilibrium (44% vs 21%, respectively) was achieved by promoting the reaction using a membrane for H2 separation in the HI decomposer, thus reducing the material flow in the decomposer by 52% (Fig.6-11(1)). To recover waste heat from the condensation of HI and iodine (I2), they were directly fed into the HI distillation column, as vapor liquid H2 separation is not needed by H2 membrane separation in the HI decomposer (Fig.6-11(2)). To recover the heat of the Bunsen reaction, previously ignored due to its low temperature (378 K), it was used in the multi-stage H2SO4 concentration flash drums by lowering boiling temperature (368 K) by depressurization with a vacuum pump (Fig.6-11(3)).
A chemical process including these technologies was designed and simulated to estimate the heat demand. Overall, the H2 production efficiency increased from 40% to 50%. Further work on the IS process components is to reduce H2 production cost based on the improved process design.