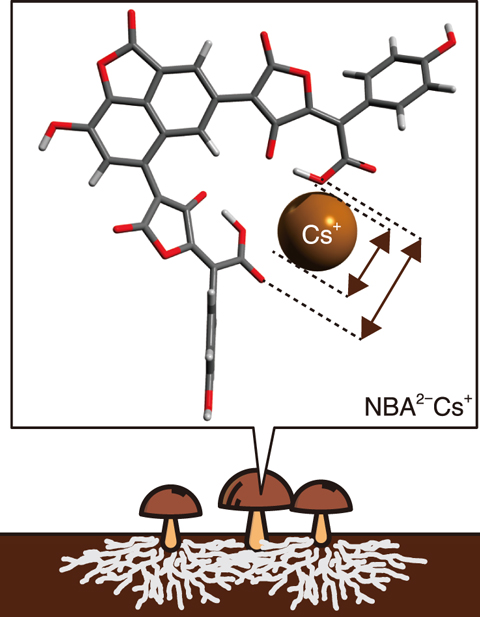
Fig.1-26 Radioactive cesium concentration mechanism in mushrooms
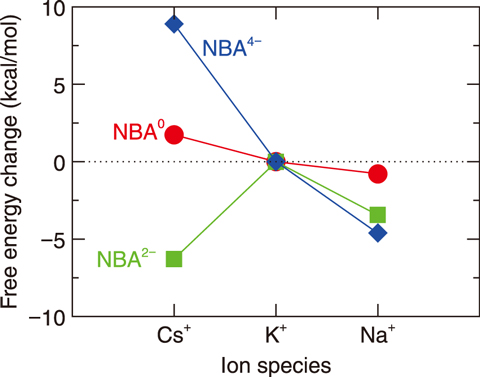
Fig.1-27 Free energy change for alkali-metal ion exchange of norbadione A in water
Radiocesium (Cs) released by the accident at the TEPCO’s Fukushima Daiichi Nuclear Power Station has been reported to be partly retained in fungi, lichens, and trees in forests, and particularly concentrated in mushrooms. The concentration of Cs in mushrooms was noticed after the Chernobyl nuclear plant accident, with high concentrations observed around the pigments contained in the mushroom cups. One representative mushroom pigment molecule is norbadione A (NBA; C35H18O15), which is the predominant pigment of the Bay Boletus in Europe and the Pisolithus arrhizus in Japan’s pine forests. Norbadione A is known to bind with Cs (see Fig.1-26), and has been investigated experimentally and via molecular dynamics simulations to clarify its selective binding mechanism. Here, the selective binding mechanism was attributed to the scissor-like molecular structure of norbadione A.
In this study, state-of-the-art quantum chemical computational techniques were used to calculate the structure of norbadione A, the free energy of formation of the binding between norbadione A and Cs, and the binding free energy binding between norbadione A and Cs, which characterizes the binding strength in a living body (i.e., in water). As potassium is generally abundant in living bodies and binds with norbadione A, the possibility of cesium concentration can be evaluated by calculating the free energy change associated with replacing a potassium ion with a Cs ion. The results demonstrated that NBA2−, norbadione A with two dissociated hydrogen ions, binds more stably with a Cs ion than with potassium or sodium ions (Fig.1-27). This indicates that cesium is selectively bound with norbadione A. For most other biomolecules, potassium binding is more stable than Cs binding, whereas Cs binding is more stable than potassium binding for norbadione A. This anomality is likely owing to the scissor-like molecular structure of norbadione A; the scissor spacing matches the cesium ion diameter, as shown in Fig.1-26. Several mushroom pigments are known to have scissor-like molecular structures such as norbadione A and badione A; their characteristic scissor-like structures may be one reason for the high concentration of Cs in mushrooms.
This research was conducted using the supercomputer SGI ICE X in the JAEA.
(Hiroya Suno)