
Fig.3-4 Snapshot of the third phase formation
Fig.3-5 Chemical structures of tri(4,4,5,5,6,6,7,7,7-nonafluoroheptyl)phosphate (TFP) and tributyl phosphate (TBP)
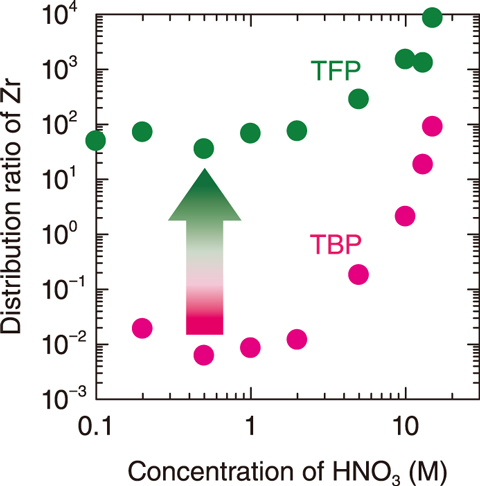
Fig.3-6 Effect of HNO3 concentration on the distribution ratio (extraction efficiency) of Zr by TFP and TBP
The efficient separation of various radionuclides is vital in reducing the amount and toxicity of radioactive waste. The separation of metal ions such as radionuclides is carried out by a hydrometallurgical process mainly based on solvent extraction, which is a separation technique used to concentrate and separate a certain target component by using distribution of a solute between two immiscible solutions. Solvent extraction has been widely adopted as a practical process in the reprocessing of spent nuclear fuel and in refining valuable metals. Further, the extraction of Zr(IV) as a fission product from aqueous HNO3 solutions has been investigated extensively because the presence of Zr in the tributyl phosphate (TBP)-based reprocessing of spent nuclear fuel is problematic. Zr species are likely to form a third phase during the extraction of Zr by TBP from HNO3 aqueous solutions, as shown in Fig.3-4.
Formation of this third phase in the reprocessing of spent fuel is undesirable, as it could cause safety and criticality concerns due to the high metal concentrations. However, few applicable extraction methods or extractants have been proposed that do not suffer from these problems. A Zr-loaded organic phase from liquid–liquid extraction with TBP was recently reported to demonstrate hierarchical aggregation behaviors of Zr(NO3)4(TBP)2 coordination complexes, which self-assemble into primary clusters that coalesce further to form superclusters. This supercluster formation portends the formation of third phase.
This work therefore aimed to develop a fluoroalkylated phosphate (TFP) for Zr(IV) extraction to increase the extraction performance and prevent third-phase formation. The structure of the developed TFP compound is shown alongside the structure of TBP in Fig.3-5. Fluorous solvents such as perfluorinated alkanes have unique chemical properties, including low toxicity, non-flammability, and immiscibility with both water and organic solutions. The high hydrophobicity of a fluorous solvent reduces the formation of a third phase and interfacial crud.
To compare the extraction capabilities of the developed TFP compound and TBP and analyze the extraction dependency on the concentration of HNO3, Zr(IV) was then extracted from an HNO3 solution by 0.05 M TFP and 0.5 M TBP; the results are compared in Fig.3-6. Despite its concentration being one-tenth of that of TBP, the 0.05 M TFP had a much higher Zr(IV) extraction performance. Moreover, no third phase formed, even as the concentration of TFP was increased. To verify the reason for the superior Zr(IV) extraction performance of TFP, the compositions of each phase before and after water, HNO3, and Zr(IV) extraction were determined using both TFP and TBP. The concentrations of water and HNO3 molecules in the fluorous phase lowered during Zr(IV) extraction, indicating that, unlike with TBP, water and HNO3 molecules do not preferentially interact with TFP. The superior performance of TFP makes it suitable as an alternative extractant to TBP in the Zr(IV) extraction system and to other conventional organic extraction systems.
(Yuki Ueda)