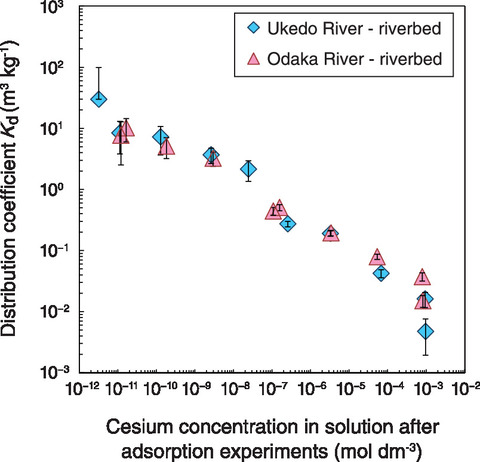
Fig.1-30 Distribution coefficient of cesium to sediments
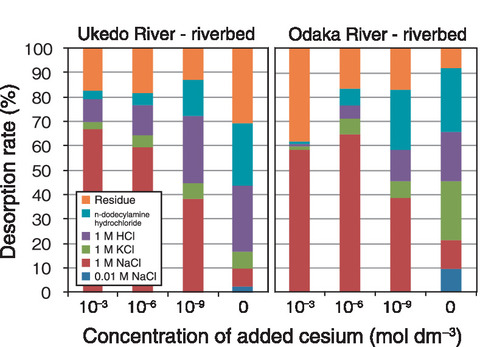
Fig.1-31 Desorption rate of cesium by sequential extraction
Radioactive cesium discharged by the accident at TEPCO's Fukushima Daiichi NPS remains in forest soil and is gradually flowing into the river system. The adsorption mechanism of cesium by sediments must be clarified to estimate the changing concentration of dissolved cesium in river water. Therefore, we collected sediments from the riverbed in the lower reaches of the Odaka and Ukedo Rivers and evaluated their cesium adsorption and desorption characteristics.
Adsorption experiments were conducted on sediments in a 0.01 mol dm-3 (M) sodium chloride (NaCl) solution with various initial cesium concentrations. The sediment samples were sieved using a pore size of 2 mm. After 68 days, when the adsorption equilibrium was attained by time dependence, the ratio of adsorbed cesium to cesium in solution (i.e., the distribution coefficient, Kd) increased in all collected river sediments as the concentration of cesium in the solution decreased (see Fig.1-30). A portion of the minerals residing in the sediment thus likely easily adsorb cesium; however, due to low site to absorb, the adsorbed rate of cesium is relatively high when the concentration is low.
Next, the sediments after the adsorption experiments were added stepwise into the solution with different desorption effects. As the concentration of initially added cesium decreased, the proportion of components adsorbed via ion exchange and then desorbed by NaCl and potassium chloride (KCl) decreased. On the other hand, the proportion of components desorbed by hydrochloric acid (HCl), which dissolves the mineral surface, and dodecylamine hydrochloride, which spreads the interlayer spacing of clay minerals, increased (Fig.1-31). We determined that the mineral in the sediment has a mixture of adsorption sites which easy to desorbed and difficult to desorbed. The Odaka River had a higher proportion of components adsorbed via ion exchange in the sediment than in the Ukedo River, indicating a higher content of smectite than mica minerals; cesium adsorption onto smectite was dominated by ion exchange.
The concentration of dissolved radioactive cesium in river water is very low (i.e., approximately 0.1 Bq/L in the Ukedo River) due to the strong adsorption of cesium onto the part of the minerals from which cesium cannot easily be desorbed. The adsorption and desorption characteristics of cesium from these minerals will aid efforts to estimate the concentration of radioactive cesium in river water.
(Kenso Fujiwara)