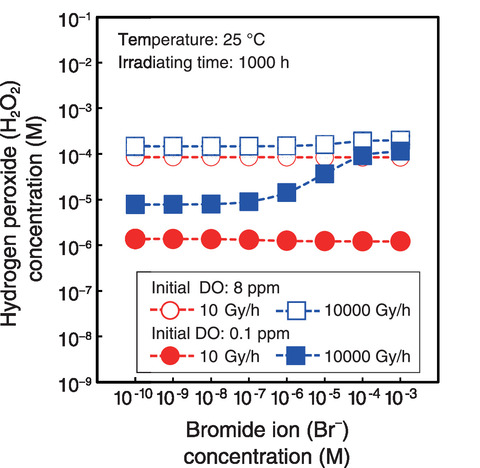
Fig.1-20 Example calculation of water quality under irradiation using the developed radiolysis database
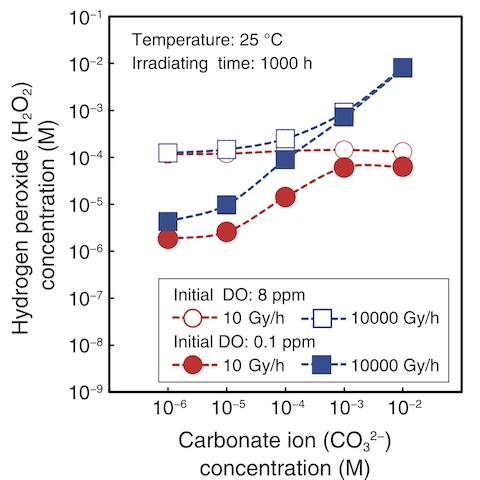
Fig.1-21 Example calculation of water quality under irradiation using the developed radiolysis database
Stagnant water and cooling water inside the Primary Containment Vessel (PCV) are exposed to radioactive materials, including fuel debris and released cesium. Further, these materials may contain many impurities, such as ions from seawater and metal ions released from steel. The PCV is currently filled with nitrogen gas. However, the PCV will likely be opened during decommissioning, allowing an inflow of oxygen (O2) gas, which will lead to variation of dissolved oxygen (DO; 0-8 ppm). Hydrogen peroxide (H2O2) accelerates the corrosion of metals and is generated by water radiolysis under irradiation. Thus, generated H2O2 may accelerate the corrosion of PCV structural materials and hinder decommissioning.
Therefore, we are developing a database that can be used to estimate the radiolysis and corrosion of structural materials due to the presence of water containing various impurities under a wide DO concentration range under irradiation. This work has involved collaborations between JAEA and the National Institutes for Quantum Science and Technology, Osaka Prefecture University, Tokyo Institute of Technology, Tohoku University, and the University of Tokyo. This database comprises three parts: a radiolysis database, a corrosion database on corrosion under irradiation, and investigation sheets on corrosion risks present during decommissioning. We at JAEA have mainly focused on building the radiolysis database.
The rate constants for 215 reactions of radiolytic species, including pure water radiolysis and chloride (Cl-), bromide (Br-), sulfate (SO42-), carbonate (CO32-), bicarbonate (HCO3-), and ferrous (Fe2+) ions, have been collected to build this radiolysis database.
The calculated H2O2 concentrations for Br- and CO32- are shown in Figs.1-20 and 1-21, respectively. Here, the corrosion rate of carbon steel (i.e., a material used in the PCV) increased with increasing H2O2 concentration. Overall, the results demonstrated that the DO, Br- ion, and CO32- ion concentrations must be reduced to reduce the generation of H2O2 under higher dose rates. The amount of radiolytic products varied by the effects of anions contained in seawater and ferrous ion released from steel on radiolysis. The developed database is expected to be applied to evaluate corrosive environments and hydrogen generation by radiolysis in containers storing contaminated water. Future efforts will include expanding the developed database to encompass a wider range of conditions and using it to analyze corrosion-prevention countermeasures.
This work was conducted as a part of the Nuclear Energy Science & Technology and Human Resource Development Project “Analysis of Corrosion Mechanism in Specific Environment” funded by the Ministry of Education, Culture, Sports, Science and Technology (MEXT), Japan.
(Tomonori Sato)