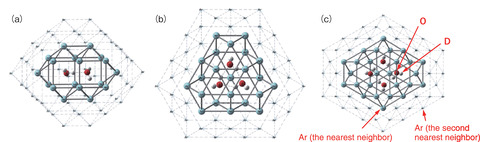
Fig.3-9 The most stable structures of deuterated water clusters trapped in solid Ar
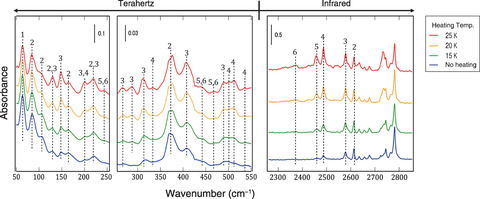
Fig.3-10 Heating-temperature dependence of the terahertz-infrared spectrum of deuterated water clusters
Nano-sized water clusters have been intensively investigated in a spectroscopic way to reveal the nature of hydrogen bond interaction. Compared with the infrared (IR) range, only a few spectral measurements in the terahertz (THz) range have been reported, so that the assignment of the vibrational mode to THz absorption was not established even for the dimer. In the present study, we measured THz-IR absorption spectra of deuterated-water (D2O) cluster formed in solid Ar and conducted ab initio vibrational calculation to identify the THz absorption peaks of the dimer, trimer, and tetramer.
A gaseous mixture of Ar and D2O was condensed on the gold plate cooled down to 8 K by liquid helium. The most stable structures (Fig.3-9) and vibrational frequencies of the dimer, trimer, and tetramer trapped in solid Ar was determined by ab initio calculation.
In Fig.3-10, THz-IR spectra before and after heating the sample are displayed. The IR-absorption peaks due to clusters larger than the dimer were found to increase in absorbance with raising the heating temperature. This means the cluster-size growth due to the thermal diffusion of D2O. By comparing the heating-temperature dependence of the peak intensities in the THz range with the IR range and using the results of ab initio calculation, we determined the assignments of the THz peaks to the vibrational modes of the dimer, trimer, and tetramer.
The above accomplishment leads to the precise description of hydrogen-bond potentials, which characterize the physicochemical properties of ice and water. In addition, the fact that the greenhouse effect of water vapor is enhanced by clustering in atmosphere has been revealed recently. The cluster-size dependence of the THz-absorption intensity revealed in the present work will play an essential role in accurate evaluation of the greenhouse effect of water, which is one of the major topics in atmospheric and environmental science.
We appreciate support from the Japan Society for the Promotion of Science (JSPS) KAKENHI Grant-in-Aid for Early-Career Scientists (No.JP18K14182).
(Koichiro Yamakawa)