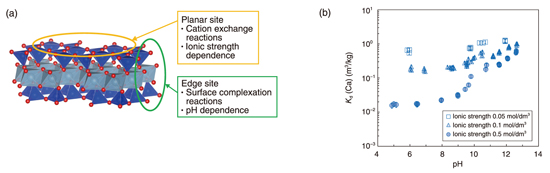
Fig.8-24 Structure of montmorillonite and sorption behavior of Ca
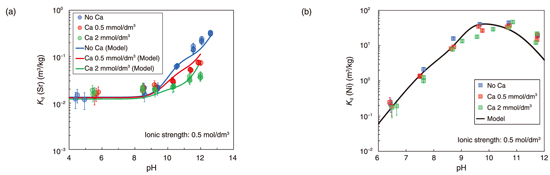
Fig.8-25 Effect of Ca on the sorption behaviors of Sr and Ni
Cementitious materials are planned to be used in high-level radioactive waste repositories. The calcium (Ca) concentration in the porewater of buffer materials will increase because of the leaching of cementitious materials. The buffer material sorbs radionuclides and retards their migration. However, the sorption of radionuclides will reduce because of the sorption competition with Ca at higher Ca concentrations. The degree of sorption of radionuclides onto the buffer material or clay minerals in rocks is expressed by the distribution coefficient (Kd; m3/kg). Although it is desirable to know Kd values under various conditions to assess the safety of geological disposal, it is not realistic to measure these values under all the possible conditions expected in a repository because the Kd values depend on pH and solution composition. Therefore, the thermodynamic sorption model, which is used to predict Kd based on the sorption mechanism, has been studied. In this study, we investigated the sorption of Ca on montmorillonite, a prominent clay mineral with regard to the sorption of radionuclides sorption in the buffer material. We also evaluated the effect of Ca on the sorption of strontium (Sr) and nickel (Ni), which are important elements for safety assessment.
In general, sorption on clay minerals can be classified into cation exchange reactions at planar sites and surface complexation reactions at edge sites. The former is affected by ionic strength and the latter by pH (Fig.8-24(a)). It is known that Ca is sorbed by cation exchange reaction. However, we found that the Kd values of Ca increased significantly at high pH under conditions of high ionic strength, and that sorption by surface complexation reactions becomes more dominant in this pH region (Fig.8-24(b)).This trend was also found for Sr, a homologous element to Ca (Fig.8-25(a)). In contrast, the Kd values of Ni increased in acidic pH region, indicating that the surface complexation reactions were dominant even at low pH (Fig.8-25(b)). Next, we performed sorption competition experiments to evaluate the effect of Ca on the sorption of Sr and Ni on the edge sites. The results indicated that Sr sorption was inhibited by the presence of Ca, while Ni sorption was not affected. This suggests that Sr sorbs at the same site a Ca, whereas Ni sorbs at a different site. Based on these results, we used a thermodynamic sorption model with a geochemical calculation code PHREEQC to reproduce the measured Kd values and derive the sorption parameters for each element. The calculations using the derived sorption parameters reproduced well the results of sorption competition experiments.
The experiments and model calculations conducted in this study clarified that elements sorb to different edge sites depending on their chemical properties. The thermodynamic sorption model can predict Kd values under various conditions arising from the long-term transition of the repository condition based on the sorption parameters obtained through various experiments.
This study was a part of “Development of enhancing the disposal system in the coastal region (FY2018)“ supported by METI (Grant number JPJ007597).
(Yuki Sugiura)