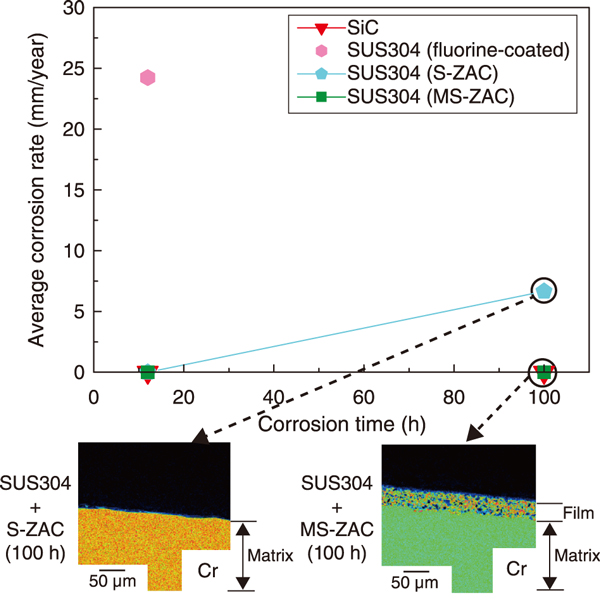
Fig.1 Change in corrosion rate with increasing corrosion time for samples, and Cr element mapping of the coating or matrix cross section in SUS304 coated with S-ZAC and MS-ZAC
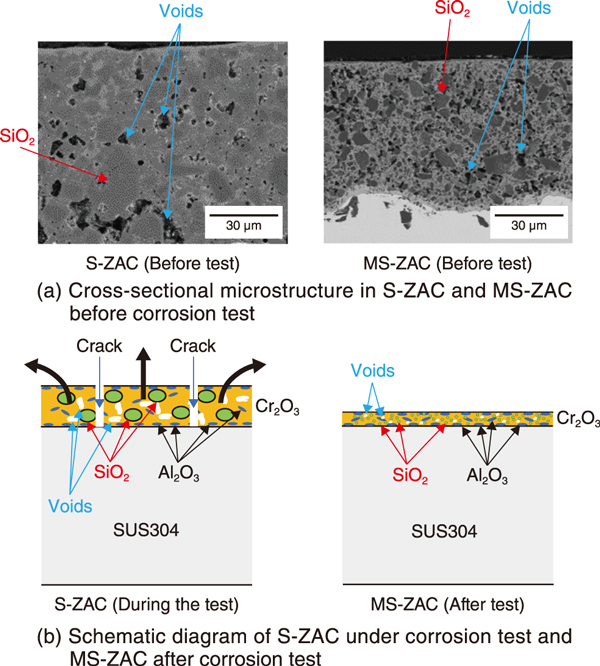
Fig.2 (a) Cross-sectional microstructures of SUS304 coated with S-ZAC and MS-ZAC before the corrosion test, and (b) schematic diagram of S-ZAC under the corrosion test and MS-ZAC after the corrosion test
The iodine-sulfur (IS) process, which is a thermochemical hydrogen production method, is a heat application technology in HTGRs. This requires structural components with excellent heat and corrosion resistance. In particular, the sulfuric acid decomposer, in which liquid sulfuric acid is transformed into a boiling state, has the most severely corrosive environment in the IS process. Therefore, SiC ceramics are yet to be used. However, SiC sintering furnaces are restricted by size limitations, and it is difficult to scale up their components for commercial production.
Therefore, we considered the development of a metallic material with corrosion resistance equivalent to that of SiC by forming a dense film on the metal surface that can be easily scaled up. Test specimens were prepared from fluorine-coated SUS304 and SUS304 coated with high-density oxide slurry. The chemical densified coating method involves coating the matrix with a slurry of Cr2O3, Al2O3, and SiO2 particles, followed by sintering at 500 ℃−600 ℃. We prepared S-ZAC, in which sintering was performed eight times, and MS-ZAC, in which sintering was performed 11 times to further densify the coating.
After the corrosion testing, the fluorine-coated material was completely delaminated. It exhibited a high corrosion rate after the 12 h test, as shown in Fig.1. Moreover, the S-ZAC-treated material maintained its corrosion resistance even after the 12 h test, but the coating peeled off after the 100 h test. However, the highly densified MS-ZAC-treated material showed corrosion resistance comparable to that of SiC after the 100 h test. Cr element mapping of the coating and matrix cross sections after 100 h tests showed that S-ZAC disappeared, whereas the MS-ZAC remained dense with fine Cr, Al, and Si particles of size 5 μm or less.
Fig.2(a) shows the cross-sectional microstructures of S-ZAC and MS-ZAC before the corrosion testing. Coarse SiO2 aggregates and voids were observed in S-ZAC, whereas finer oxide particles, fewer voids, and thinner films were observed in MS-ZAC (Fig.2(b)).
Therefore, considering the condition of the film in the boiling sulfuric acid test environment, it is speculated that in S-ZAC, the Cr2O3 surrounding the SiO2 began expanding, and tensile stress acted at the Cr2O3-SiO2 interface, causing the thick film to curve, leading to film delamination. In contrast, MS-ZAC maintained a thin film because the SiO2 particles became finer during the impregnation process, resulting in smaller voids. In the future, we plan to study the thickening of the MS-ZAC films.
This study is a part of the collaborative research with TOCALO Co., Ltd., on “Research on New Coating Materials Improving Corrosion Resistance Under IS Process Environment.”
(Noriaki Hirota)
<Previous: 1-6 | Next: 2 Research and Development of Fast Reactors>