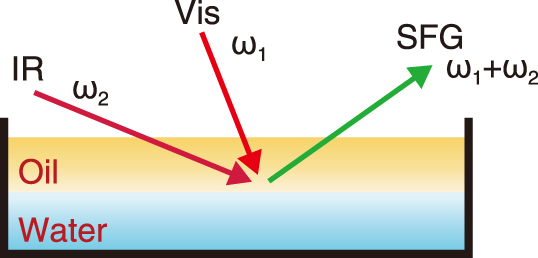
Fig.1 Vibrational sum frequency generation (VSFG) spectroscopy measurement
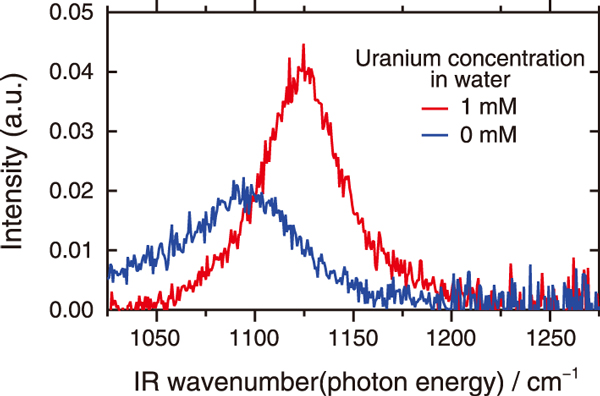
Fig.2 VSFG spectral data
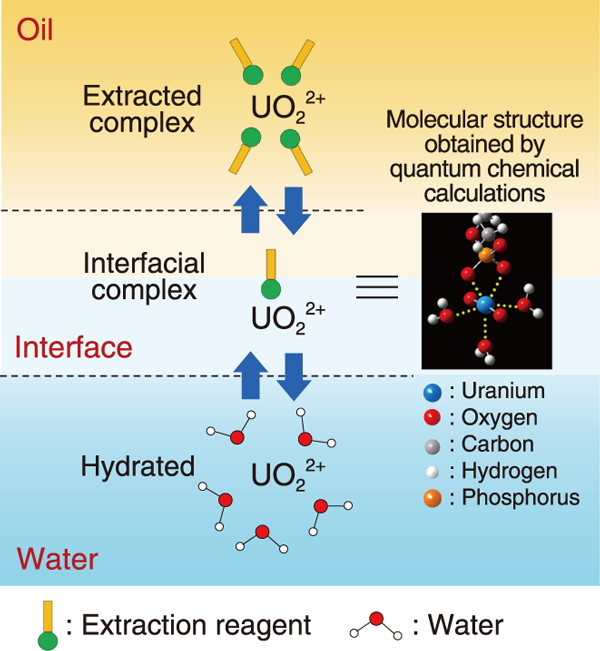
Fig.3 Model of uranium phase-transfer reaction
Vibrational sum frequency generation (VSFG) spectroscopy is an advanced laser spectroscopy method that provides insights into the molecular structure at boundary regions (interfaces) where two different phases are in contact. As yet, there is a lack of research institutions capable of measuring nuclear fuel materials and radioactive isotopes using VSFG spectroscopy. In this study, we built a VSFG spectroscopy apparatus within our radiation-controlled area. Using this setup, we examined the mechanism of uranium transfer between the water and oil phases at the oil-water interface of the solvent extraction separation method for uranium.
In the VSFG spectroscopy measurement (Fig.1), visible and infrared lights were incident on the oil-water interface, and the light generated from the interface (i.e., sum-frequency light) was detected. Fig.2 shows the VSFG spectroscopy data obtained. When uranium was dissolved in either the water phase or the oil phase, the signal peak position shifted from 1100 cm-1 (![]() ) to 1125 cm-1 (
) to 1125 cm-1 (![]() ). Analysis using the quantum chemical calculation program (Gaussian) confirmed that this shift can be attributed to the binding of two oxygen atoms of the POO- group in the extraction reagent to the uranium atom of the uranyl ion (UO22+) at the interface (Fig.3).
). Analysis using the quantum chemical calculation program (Gaussian) confirmed that this shift can be attributed to the binding of two oxygen atoms of the POO- group in the extraction reagent to the uranium atom of the uranyl ion (UO22+) at the interface (Fig.3).
It is well-known that uranium exists in different chemical states in the two phases−it exists as hydrated ions in the aqueous phase and as extraction complexes in the organic phase (Fig.3). However, the mechanism of how uranium passes through the interface is unknown. The findings of this study indicate that uranium moves between the two phases by forming a molecular structure as shown in Fig.3 with the extraction reagent at the interface.
Evidently, uranium has to pass through the oil-water interface to complete the extraction process, implying that the interface is the crucial reaction zone in solvent extraction. Further elucidation of the reactions occurring at the interface has potential for promoting the development of innovative uranium solvent extraction methods by using interfaces.
This research was supported by JSPS KAKENHI Grants-in-Aid for Young Scientists (B) (JP17K14919) and Grants-in-Aid for Scientific Research (C) (JP20K05388).
(Ryoji Kusaka)