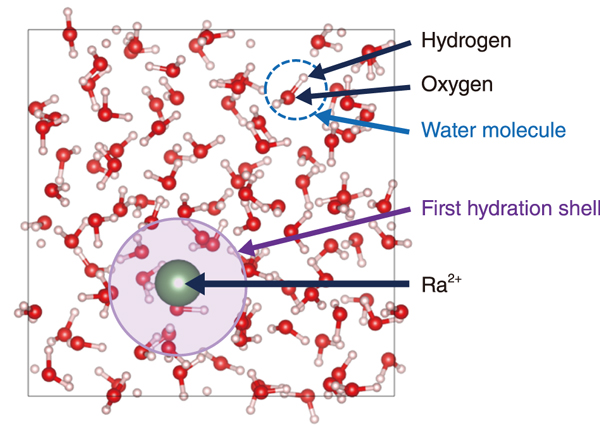
Fig.1 Snapshot of hydrated Ra2+ simulation
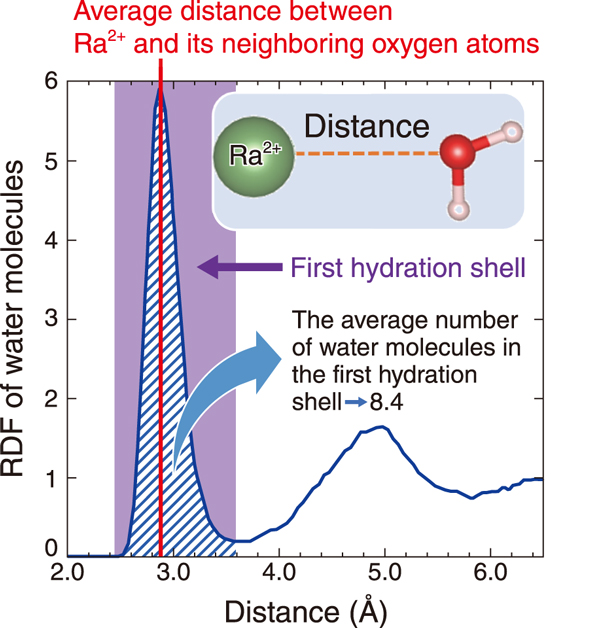
Fig.2 Distribution of water molecules hydrating Ra2+
Table 1 Comparison of experiment and simulation results

Radium (Ra), discovered by Marie and Pierre Curie in 1898, is now attracting attention in diverse fields such as medicine and earth sciences; meanwhile, environmental pollution by Ra is becoming a concern. However, details of Ra chemical reactions in living organisms and environment remain unknown. Since these reactions primarily occur in water, the structure of Ra dissolved in water (i.e., hydrated Ra2+) is the most fundamental and important information. However, since Ra is a radioactive element without a stable isotope, and radon, a gaseous radioactive element, is produced by Ra decay, there is high risk of internal exposure during its study, and a high level of safety control is required for Ra experimentation. Therefore, >100 years after the discovery, no experiments at the molecular level have been conducted on this element.
In this study, we developed a container that prevents radon leakage and established a method to safely prepare, transport, and measure high-concentration Ra samples. Then, using SPring-8, we successfully performed the first molecular-level measurement of hydrated Ra2+. In addition, ab initio molecular dynamics simulations were performed using a supercomputer (Fig.1). The results (Fig.2) successfully reproduced the experimental results (Table 1). A more detailed analysis of the simulation results and comparison with barium (Ba), an element similar to Ra, revealed that the average residence time of water molecules in the first hydration sphere of Ra2+ (38 ps) is shorter than Ba2+ (98 ps), indicating that Ra2+ has a weak binding with the hydrating water molecules. The results indicate that the hydration structure of Ra2+ can be changed more easily than Ba2+, suggesting that compared to other alkaline earth metals, Ra2+ is more likely to leave water and be incorporated into the solid phase in living organisms and the environment.
To summarize, in this study, we established a method for studying Ra chemistry at the molecular level using synchrotron radiation experiments and simulations. In the future, this method is expected to be applied to study more complex chemical reactions and may help us understand the mechanisms of cancer drugs; additionally, it is expected to make possible novel drug development, improved soil age estimation techniques, and resolution of environmental concerns and other important social problems.
This study was conducted in collaboration with the University of Tokyo and Osaka University, and supported by JSPS KAKENHI Grant-in-Aid for Research Activity Start-up (JP19K23432) and by JAEA Fund for Exploratory Researches (Houga fund).
(Akiko Yamaguchi)