A polymer gel is composed of macro-molecules which are crosslinked chemically or physically. The 3-dimensional mesh thus formed becomes greatly swollen, incorporating a high concentration of small solvent molecules. Gel-based materials occur frequently in living tissues, e. g., the cornea of the human eye. Softness is one of the most striking characteristics of a polymer gel. At the same time, unlike a liquid, it never flows. Gels possess the contrary aspects of softness and non-fluidity, which suit them for use in living tissue.
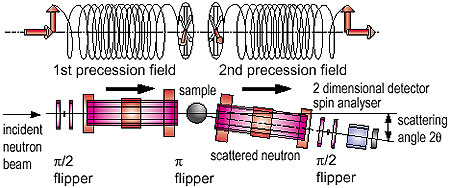
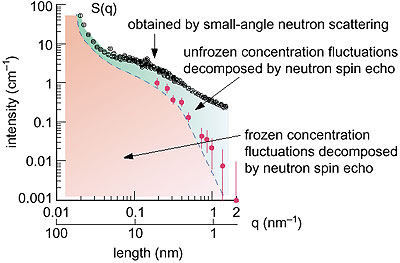
What is the origin of these sharply contrasting characteristics? Small-angle Neutron scattering and neutron spin echo spectroscopy offer microscopic probes to examine this question. If we observe a polymer gel on length scales smaller than the mesh size, we find no difference between a gel and a normal polymer solution. Accordingly, thermally excited concentration fluctuations are free from any internal bonding effects. However, if the length scale of observation becomes larger, comparable with a mesh size, this picture is no longer valid. The internal crosslinks behave like pinning to freeze part of the concentration fluctuations in the gel and induce a permanent elastic modulus. Thus, there are two different length scales of concentration fluctuations, i.e., frozen and unfrozen. This crossover, which happens on nano-length scales, is at the origin of the two contrary aspects of gels. Furthermore, the pinning in a gel is strongly affected by thermal concentration fluctuations. Therefore, the spatial distribution of pinning is very sensitive to temperature, which is quite different from that which occurs in a solid. |