In radioactive pollution, the most important nuclei are radioactive isotopes of iodine, which are volatile and easily accumulated in the thyroid gland. The radioactive isotopes change to stable nuclei immediately, and they are completely indistingishable from nuclei which exist in the environment. This makes the quantification of radioactive pollution difficult in the case of nuclear accidents, etc.
Since radioactive 129I has a half-life as long as 15.7 million years, it can be used as an index of environmental pollution relative to the radioactive isotopes 131I or 133I. This is used as an index not only for short-life radioactive nuclear pollution caused by accidents, etc., but also for long-term contamination brought about by nuclear testing or fuel reprocessing. However, since the quantity of 129I is very low in general, it is very difficult to distinguish the latter sources from radioactivity which occurs naturally in the environment. Up to now, 129I has been quantified by removing the contaminating nuclide using radiochemistry techniques, involving unavoidably complicated procedures which entail a great deal of exposure to workers. Therefore, a practical quantitative analysis with high sensitivity has long been sought after.
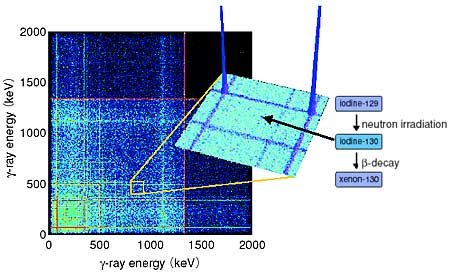
Neutron activation analysis utilizing multiple gamma-ray detection is a technique based on gamma-ray coincidence methods, and was developed at the Department of Materials Science. It has achieved about 1000 times more accurate quantification than neutron activation analysis using a single conventional detector because of the synergistic effect of resolution using two or more gamma-ray detectors. The quantification of 129I has been performed using an array of 12 germanium detectors with Compton suppressors, GEMINI (Fig. 6-1). A very low detection limit of 10-13 for the ratio of 129I to stable 127I was achieved without using radiochemistry techniques (Fig. 6-2). The ratio of 129I to stable 127I in seaweed was found to be 3.5x10-10 in the sea close to Tokai-mura. This level is comparable with that of the kelp from Hokkaido, and is also close to the natural background.
Using the long half-life of 129I, age measurements on a time scale of 10 million years become a practical possibility for samples such as minerals or methane hydrate from alternative energy sources which deposit on the seabed. In the fields of earth science and resource development, both scientific and industrial applications are expected. |