| In recent years the use of urania-gadolinia fuels in light water
reactors has been promoted significantly because of the social
demand for higher-burnup fuels. The U1-yGdyO2+x fuels containing several mol% Gd (y<0.1) as the burnable-neutron poison are very attractive for improving the power mismatch between fresh and partially burned fuel assemblies. Many other rare-earth elements (La, Nd, Sm, Ce, Pr, etc.) and their homologues Y are the major fission products resulting from the irradiation of UO2 fuel. These rare-earth elements similar to Gd dissolve in the
host UO2 lattice and affect significantly the high temperature irradiation
behavior of UO2 fuel such as the pellet-clad interaction, the release of fission
products, the change of the oxygen potential g (O2) under the burnup and so on. The irradiation behavior is also
crucially influenced by the initial values of g (O2) and the composition of oxygen (x) in the starting fuel. Thus, precise oxygen potential g (O2) data for the U1-yGdyO2+x system are indispensable for a better understanding and for controlling
not only the fabrication process but also the irradiation process
of the fuel. Yet, the experimental data for the U1-yGdyO2+x system are still fragmented and partly inconsistent, in contrast
to the base material UO2+x for which very extensive and thorough experimental data are available.
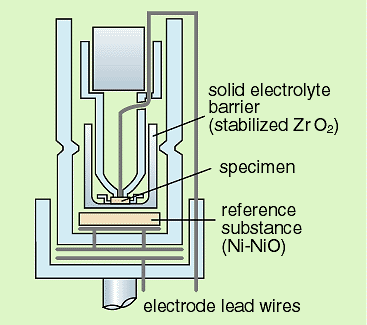
Using the solid state electromotive force (EMF) measurement as
shown in Fig. 3-1, new concise thermodynamic quantities of the
U1-yGdyO2+x have been obtained for wide ranges of the non-stoichiometric
composition of oxygen (x), the content of Gd (y) and temperature (T) (
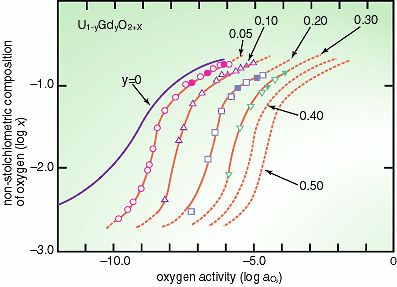
1,500 degrees). One of the experimental results is given in Fig. 3-2.
The oxygen potential g(O2), i.e. oxygen activity (aO2), steadily shifts to higher values with increasing content of
Gd (y). This information on thermodynamics is very useful to analyze
the behavior of the U1-yGdyO2+x fuels under a high-burnup and high temperature condition. |