
Fig.12-19 AMS at the Mutsu Establishment
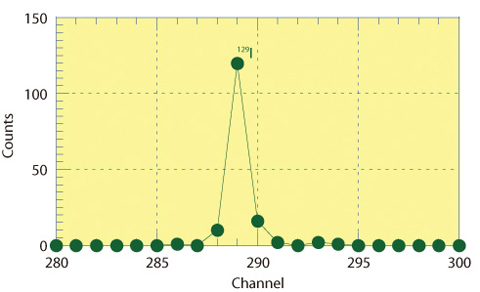
Fig.12-20 129I spectrum detected by time of flight detector
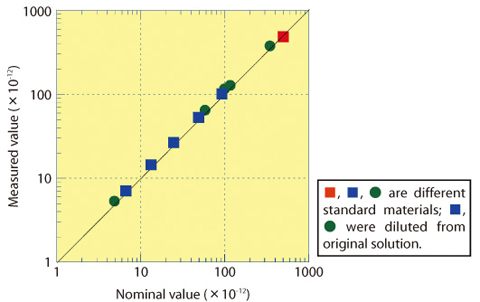
Fig.12-21 Linearity experiment
Iodine-129 (129I) is a long-lived radioactive isotope with a half-life of 15.7 million years which is released from spent nuclear fuel reprocessing plants. 129I is an important nuclide for monitoring around nuclear facilities and also useful for tracing in hydrogeologic and oceanographic research. NAA is recommended as a measurement technique for 129I in the "Analytical method of radioactive iodine-129" published by the Ministry of Education, Culture, Sports, Science and Technology of Japan. Because NAA has a high detection limit (129I/127I = 10-9-10-10), it takes much time for analysis, has low precision, and would cause radiation exposure during analysis, it is not suitable for environmental samples (129I/127I = 10-10-10-12) except for the monitoring around nuclear facilities. Therefore, a measurement technique for 129I with short processing time, high precision and high-sensitivity was developed using accelerator mass spectroscopy (AMS) (Fig. 12-19) set up at Mutsu establishment.
In mass analyzing for 129I5+ (m/e=25.8),103Rhodium4+(m/e=25.75) and 52Chromium2+ (m/e= 26.0) interfere with analysis because the mass to charge ratio is close. We succeeded in removing the interfering ions by the improvement of the mass resolution using a high mass resolution magnet, high energy resolution electrostatic deflector and time of flight detector (Fig.12-20). Also, a stable beam from the target is obtained by mixing sufficient Niobium in the target to increase conductivity, resulting in reliable measurement.
Standard samples which had a variety of iodine isotopic ratios between 10-10 and 10-12 were measured for about 60 min and the excellent linearity of the plot of nominal and measured values (Fig.12-21) shows that this AMS has good precision. Evaluating the detection limit of this method using commercial silver iodide, it was possible to measure as low as a 10-14 iodine isotopic ratio.
This measurement technique enables not only the simplification of the monitoring around the nuclear facilities but also breakthrough use of 129I in environmental migration research.