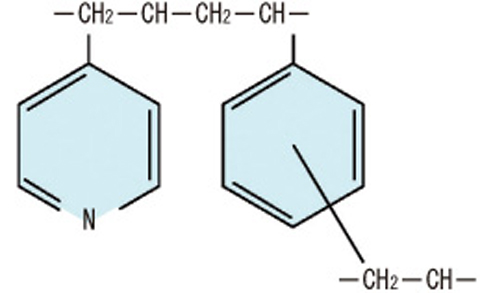
Fig.1-39 Basic structure of tertiary pyridine resin
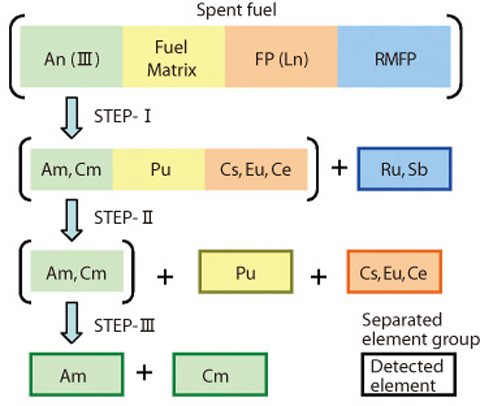
Fig.1-40 Outline of separation process
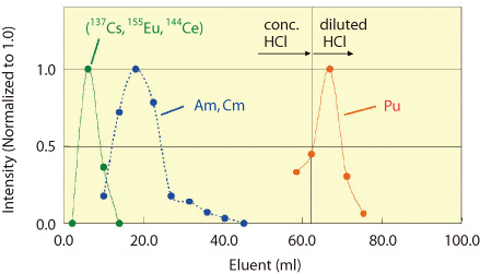
Fig.1-41 Chromatogram of STEP-II separation process
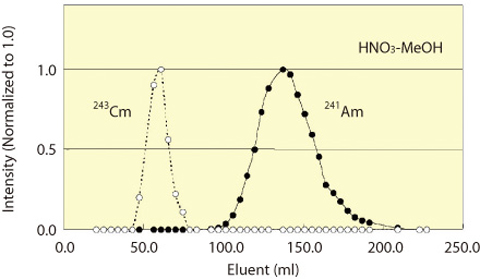
Fig.1-42 Chromatogram of STEP-III separation process
A separation process concept utilizing tertiary pyridine resin was applied to the separation of rare metal fission products (RMFP), trivalent lanthanides (Ln(III)), trivalent actinides (An(III)) and plutonium (Pu) in mixed oxide (MOX) fuel irradiated at "JOYO". Further, the mutual separation of americium (Am) and curium (Cm) was performed successfully. The study was carried out with Tokyo Institute of Technology.
The resin has the functions of a weakly basic anion-exchanger and a nitrogen soft donor ligand, and is a salt-free CHON compounds (Fig.1-39).
The separation process (Fig.1-40) can be divided as follows:
STEP-I : Platinum group elements separation (pre-filtration)
STEP-II : Separation of Ln(III) and other FP, An(III), and Pu
STEP-III : Separation of Am and Cm
Firstly, a 85μg HCl solution of MOX with high burnup of 140 GWd/MTM was introduced to the separation process.
STEP-I Antimony-125 and RMFP, mainly 106Ru in present experiment, were perfectly adsorbed in the resin. STEP-I The solvent was changed to conc. HCl solution, and passed through the resin embedded in silica beads. The Ln (III) including the other FP, such as 155Eu, 144Ce, and 137Cs, were separated. Accordingly An(III) were separated. After the removal of An(III), the effluent was changed to diluted HCl to strip out Pu fraction from the resin. (Fig.1-41) STEP-III An(III) solution was changed to nitric acid-methanol mixed solution, and passed through the resin, separating Am and Cm. (Fig.1-42)
The recovery of 241Am was more than 95% in this experiment. The Decontamination Factors (DFs) of 137 Cs and 155Eu in the isolated Am-fraction exceeded 3.9×104 and 1.0×105, respectively. The DF of 243Cm from 241Am was more than 2.2×103. This DF of An(III) is higher than ever achieved. These results proved that a simplified separation process can be used in the advanced ORIENT multifunction cycle with enhanced separation, transmutation and utilization of spent fuel.