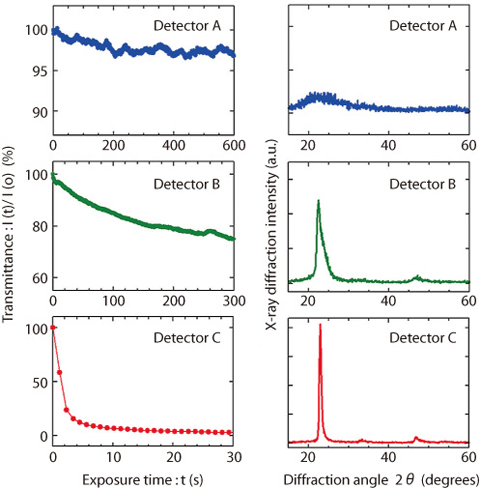
Fig.4-8 Response for hydrogen and structure of the tungsten oxide in the detectors
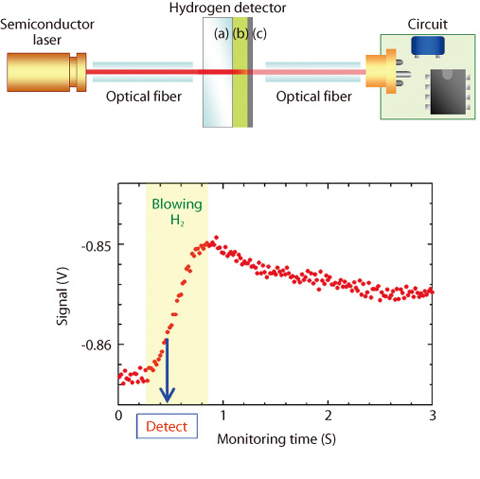
Fig.4-9 Developed optical hydrogen sensor
The concentration of lower explosive limit (LEL) in hydrogen (H2) is 4 vol.% at room temperature in ambient air. Therefore, the development of technology to detect a leak of H2 with low cost, but reliably and safely, is required for the production, the storage, the transport, and the use of H2. Optical hydrogen sensors without an electric current and a heating in the detector have been proposed as safer and cheaper sensors. The detector consists of transparent substrate, a coloration layer of tungsten oxide thin film, and catalyst layer of palladium. Leakage of H2 is detected by decrease in the intensity of transmitted light from the detector, using the gasochromic phenomenon that tungsten oxide changes color from yellow to blue when it adsorbs H2 catalyzed by palladium. The detection time is required to be the less than 1 second in the standard which is proposed by U.S. Department of Energy (DOE). But the coloration rate of tungsten oxide for H2 is usually slow, on the order of minutes. In this study, the tungsten oxide thin films with rapid coloration rate were investigated. It was found that the response in the detector using oriented crystal tungsten oxide is faster, as shown in Fig.4-8. A sensor was constructed with the detector inserted between conventional optical fibers. It was verified that the sensor can detect 1 % H2 in ambient air within 1 second, as shown in Fig.4-9. Using the optical fiber networks, the sensor can be applied to a centralized monitoring system for large pipe layouts or complex plants of production and storages.