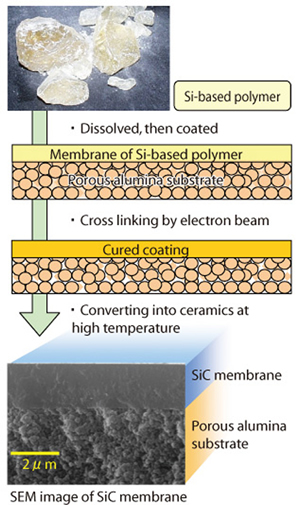
Fig.4-5 Fabrication process of silicon carbide (SiC) membrane
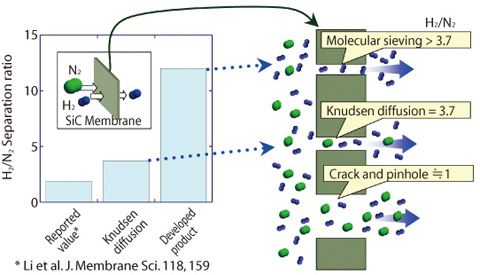
Fig.4-6 Gas separation ratio and separation mechanism of the SiC membrane
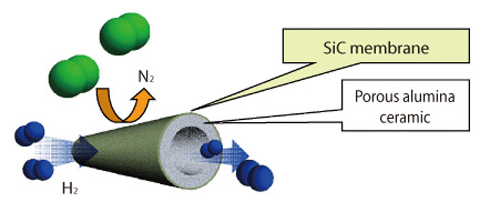
Fig.4-7 Tubular hydrogen separation membrane with SiC coating
To develop into a society that doesn't exhaust greenhouse effect gases, we are researching and developing the hydrogen fuel cell and a new thermo chemical process of hydrogen production using nuclear energy. For the fuel cell, removal of impurities from hydrogen is necessary. In the latter process, hydrogen is separated in a strong acid at over 500°C, and therefore the separation membrane material should have heat and corrosion resistance, stable for several thousand hours. Ceramics materials are the best for such applications.
JAEA succeeded in the development of silicon carbide (SiC) ceramic fiber that has high strength and heat resistance over 1700°C, by the method of converting Si-based polymer fiber to ceramics by firing after electron beam irradiation for cross linking, which prevents melting. Further, we found that there is amorphous area in that SiC ceramics, with an atomic network in which nano-holes are formed. The Knudsen diffusion mechanism controls separation of gases when both the smaller and the bigger gas molecules can penetrate through the nano hole membrane. In such case, H2/N2 separation ratio is 3.7. On the other hand, it is possible to reduce the diameter of nano holes to the size that promotes permeation of only hydrogen having the smaller molecules. This hydrogen separation membrane that functions based on the molecular sieve mechanism has high separation ratio.
Now, we are advancing the development of the SiC membrane by the fabrication process shown in Fig.4-5. In this method, it was difficult to decrease the crack formation because of the volume of Si-based polymer shrank over 50% during conversion of Si-based polymer into ceramics by firing. The volume shrinkage was reduced by electron beam crosslinking which increases the ceramic conversion rate. In addition, the formation of pinholes was lowered by mixing two kinds of Si-based polymer, to improve precursor fluidity.
Fig.4-6 shows the separation mechanism of hydrogen and nitrogen by the SiC membrane made by this method. The separation ratio of the SiC membrane developed in this research is larger than that fabricated by thermal oxidation and the theoretical value of perm-selectivity controlled by the Knudsen diffusion mechanism. In the future, we plan to improve hydrogen permeability by increasing the number of nano holes of the optimal size and the development of the cylindrical SiC membranes to produce the tilter module as shown in Fig.4-7.