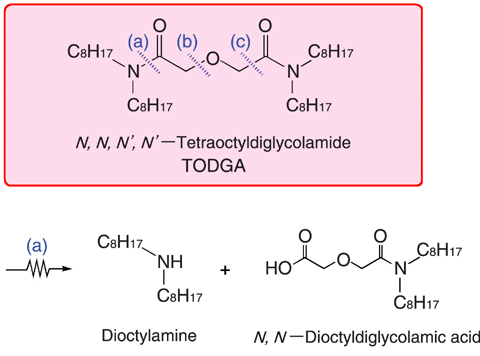
Fig.7-10 Main reaction of TODGA radiolysis
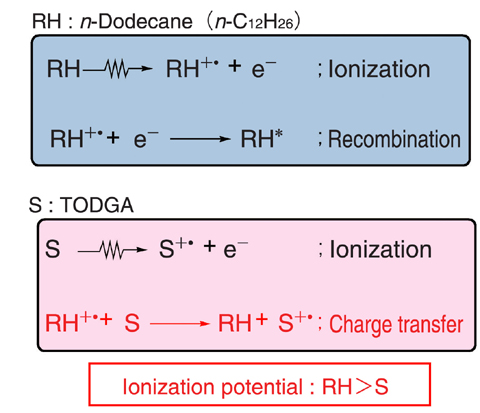
Fig.7-11 Reaction mechanism of TODGA radiolysis in n-dodecane
For the purpose of reduction in the long-term environmental burden of radioactive waste, we have developed an efficient extractant for the recovery of long-lived minor actinides from spent nuclear fuel. A tridentate diamide, N,N,N',N' - tetraoctyldiglycolamide (TODGA) efficiently extracts tri- and tetra-valent actinides from nitric acid into n-dodecane at high acidity.
In order to evaluate the applicability of TODGA to the extracting process in high radiation fields, we investigated the radiolysis of TODGA and its effect on the extraction of actinides. We also attempted to develop an improved amidic extractant, based on the reaction mechanism of the radiolysis of TODGA in the solution.
Fig.7-10 shows the main reaction of the radiolytic degradation of TODGA by γ-rays. It was found that amide- bonds and bonds in the vicinity of ether oxygen were relatively weak against radiation. In particular, in the presence of nitric acid, the cleavage of the amide-bond (a) is dominant, and mainly dioctylamine and N,N-dioctyldiglycolamic acid are formed. Furthermore, the radiation effect on the extraction of actinides was investigated using a solution of TODGA pre-irradiated with γ-rays. Good extraction at high acidity was maintained even after irradiation up to a dose corresponding to dozens of cycles in the actual process. This result suggests that there are some radiolytic degradation products contributing to the extraction of actinides.
Next, the radiolytic stability of TODGA in n-dodecane containing various additives was investigated. Since n-dodecane has a sensitization effect on the radiolysis of TODGA in the solution, reduction of the mole fraction of n-dodecane by the addition of benzene or N,N-dioctylhexanamide protects TODGA from radiation.
From our pulse radiolysis study, we concluded that the sensitization effect of n-dodecane was mainly due to a charge transfer reaction from the radical cation of n-dodecane (RH+.) to the TODGA molecule (S) as shown in Fig.7-11. The ionization potential of TODGA is less than that of n-dodecane, and the difference in the ionization potentials brings about the charge transfer reaction. Accordingly, by using solvents whose ionization potentials are lower than TODGA, TODGA can be protected from radiation.
In addition, it was confirmed that the radiolytic stability of diamide was effectively improved by the substitution of alkyl groups with aromatic groups, which are much more resistant to radiation damage. A still more improved extractant, which has high radiolytic stability as well as high ability to extract actinides, is being developed.