Fig.14-6 Steps in U-Pu cocrystallization reprocessing
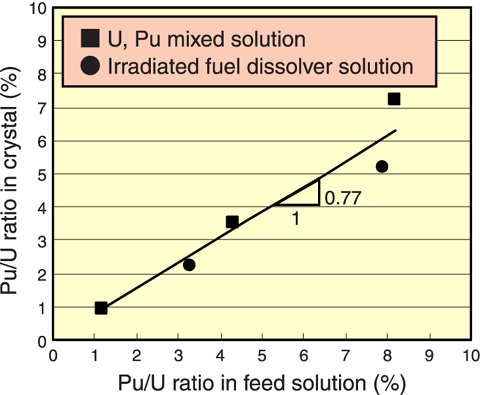
Fig.14-7 Comparison between Pu/U ratio in feed solution and Pu/U ratio in crystal
Crystallization is considered to be one of the most promising methods for recovering uranium (U) and/or plutonium (Pu) in next-generation reprocessing. The principle of this method is based on the different temperature dependency of U and Pu solubility, which means no special reagent or extractant is required. Using U and Pu mixed solutions obtained by dissolution of irradiated fuel from the experimental fast reactor "JOYO" or unirradiated MOX (mixed oxide of U and Pu), fundamental experiments have been conducted in the hot laboratory named CPF (Chemical Processing Facility).
Through experiments, it has been revealed that hexavalent Pu (Pu(VI)) was co-crystallized with U even though Pu concentration was below its solubility limit. Taking advantage of this phenomenon, an innovative reprocessing process relying on crystallization alone was created (Fig.14-6), which features are;
・Pure Pu cannot be recovered.
・U/Pu ratio in the U, Pu mixed product is controllable by means of only U recovery posterior to the co-crystallization.
・Organic reagents are not used
The mechanism of U/Pu co-crystallization is considered to be similar to co-precipitation where the target element reacts with a carrier compound and a reagent additive. Usually, the carrier has a crystalline structure similar to that of the target element. The nitrate hexahydrates of U and Pu have the same type of crystal structure.
In the U-Pu co-crystallization, Pu is the target element and U acts as the carrier. U crystal is produced by temperature dropping instead of reagent addition, and Pu precipitates together with it. Conducting experiments using solutions with several kinds of U/Pu ratio, it has been confirmed that the U and Pu were co-crystallized under any conditions. It has also become clear that the U/Pu ratio in the crystal is lower than that of the feed solution (Fig.14-7).
In order to commercialize this system, methods for improvement of U/Pu co-crystallization yield and purification of resulting U and Pu must be developed.
This work was financed by the Ministry of Economy Trade and Industry of Japan (METI).