
Fig.14-6 Plutonium cesium nitrate obtained from a nitrate solution
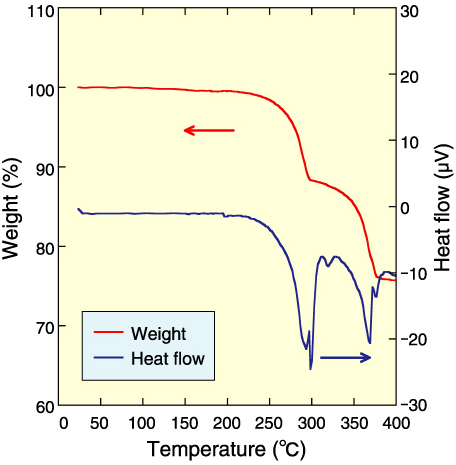
Fig.14-7 Thermal analysis of plutonium cesium nitrate
Crystallization has been studied as advanced aqueous reprocessing in order to partly separate uranium from the dissolver solution of fast breeder reactor (FBR) spent fuel. Crystallization is applied in the fields of chemicals, food, and medicine. In the 1980s, uranium crystallization was proposed by the Kernforschungszentrum Karlsruhe (KfK) in Germany for purifying the uranium product in light water reactor (LWR) fuel reprocessing. Uranyl nitrate hexahydrate (UNH) can be separated and recovered only by temperature control of the solution without a further reagent. Since the dissolver solution of FBR spent fuel contains more plutonium and fission products (FPs) than the uranium product obtained from solvent extraction by the KfK, it is important to understand the behavior of these elements. Previous studies have suggested that plutonium cesium nitrate precipitates in the uranium crystallization, and there may be issues induced by low purified UNH crystals and increasing radiation exposure during uranium product storage and fuel fabrication. Therefore, we studied the physicochemical properties of plutonium cesium nitrate in order to suppress their precipitation and removal from the UNH crystal.
Precipitation experiments were carried out at the Chemical Processing Facility (CPF). Pale green precipitate was prepared from a nitrate solution (Fig.14-6). This compound has been proven to be generated with increasing HNO3 concentration in the mother liquor. This compound was identified Cs2Pu(NO3)6 by elemental analysis and X-ray diffraction. The thermal properties of Cs2Pu(NO3)6 were studied for purification operations at 60~100 °C. Since Cs2Pu(NO3)6 weight loss was not observed below 245 °C (Fig.14-7), the compound was shown to exist in a solid state during the purification operation.
The Cs2Pu(NO3)6 experimental data will be useful for finding a way to suppress its precipitation and to recover highly purified UNH crystals.
This work was financed by the Ministry of Education, Culture, Sports, Science and Technology of Japan (MEXT).