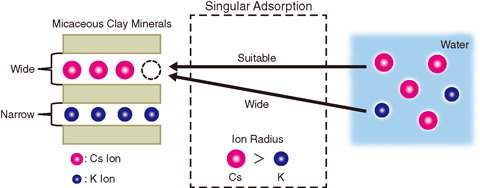
Fig.1-17 Model of ion-exchange reaction between micaceous clay mineral and water and the mechanism of singular adsorption
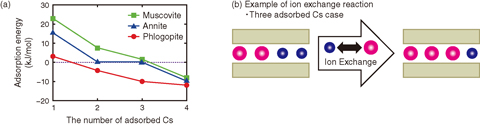
Fig.1-18 Cs adsorption energy of micaceous clay minerals
The accident at the TEPCO’s Fukushima Daiichi NPS in 2011 released many radioactive nuclides into the environment. In particular, radioactive cesium (Cs) has been the main γ-ray emitter because it has a long half-life and has been retained by surface soil. The Japanese government has performed large-scale decontamination, which has reduced radiation. However, a new issue arose: treatment of huge amounts of waste soil.
It is known that Cs in soils is adsorbed by micaceous clay minerals. The waste volume can be reduced if the Cs is desorbed from them. However, there is no economical or effective technique for this reduction owing to lack of knowledge about adsorption sites at an atomic scale. Therefore, it is necessary to reveal the mechanism of the strong adsorption of Cs to the micaceous clay minerals for development of the desorption techniques.
It is expected that Cs released into the environment would have been dissolved into water as an ion and then adsorbed to micaceous clay minerals by ion-exchange reaction with potassium (K) ions preoccupying the clay minerals. Recently, a singular Cs adsorption to a clay mineral has been observed: Cs ions are accumulated by biotite.
Therefore, we made models of muscovite, phlogopite, and annite, which are end members of biotite, on a supercomputer (Fig.1-17). We simulated ion-exchange reactions and evaluated their energy using a first-principles calculation (Fig.1-18). We obtained the result that serial adsorption increases the affinity of clay minerals for Cs. In particular, phlogopite and annite show stronger affinity than muscovite. In addition, this tendency is explained by a simple model: matching of ion radius and interlayer distance (Fig.1-17). This result shows that the singular adsorption observed is a general phenomenon in the environment. We continue our study to develop a technique for waste volume-reduction, which contributes to the reduction of environmental damage.