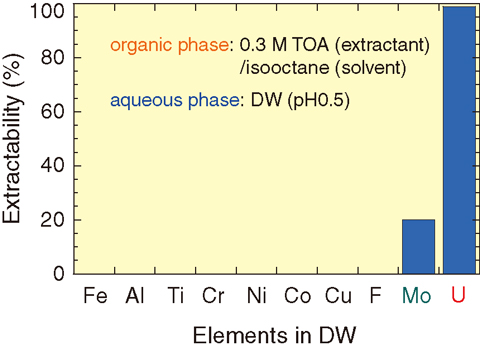
Fig.4-15 Selective extraction of U from decontamination wastewater

Fig.4-16 Recovery of suspended particulate matter in decontamination wastewater

Fig.4-17 Removal experiment for U in decontamination wastewater
At the Ningyo-toge Environmental Engineering Center, a large quantity of uranium-containing wastewater has been generated from the decontamination of facilities or apparatuses. The decontamination wastewater (DW) is treated by coagulation-precipitation processes to form radioactive sludge, into which the uranium (U) in DW passes. A selective removal of U from DW is required to reduce the U content of the sludge and to dispose it of more easily. Uranium-removal methods using ion exchange resin or chelate resin have been previously considered, but they have challenges of low processing speed and high cost.
We here introduce a simple, safe, and low-cost liquid-liquid extraction method using a newly developed “emulsion flow” (EF) extractor, wherein a flow of emulsified phases, referred to as EF, is originated by spraying fine droplets of extractant-containing organic phases into aqueous phases. The distinguishing features of the EF method are high extractability due to sufficient mixing of the organic and aqueous phases and high processing speed due to the high separation ability of the emulsified phases in the EF apparatus. Using an adequate extractant, U can be selectively extracted from DW that contains various metal ions (Fig.4-15). The EF method is also applied to collecting and removing suspended solids (SS) by accumulating them at a water/oil interface (Fig.4-16). The use of the EF method allows the treatment of DW with higher speed (more than tenfold) and higher efficiency at a low cost (less than one fifth) compared with conventional techniques using ion exchange resin and the like.
An experiment using an EF extractor with about half the size of the practical scale (Fig.4-17) was conducted to treat actual DW generated at the Ningyo-toge Environmental Engineering Center with a speed of 60-90 ℓ/h. As a result, we succeeded in promptly removing U in DW (3 Bq/cm3) below the effluent standard level (0.0022 Bq/cm3) and in recovering SS in the wastewaters into the recovery part of the extractor. It was also found that connecting three EF extractors would permit the selective removal of more than 99.9% of U from DW.
The EF method is expected to be widely applicable for the treatment of domestic uranium-containing wastewaters. This new technology has received much attention not only in the nuclear field but also in many industrial fields because of its usefulness for purifying industrial wastewaters or recovering rare metals from them.
This study received the 47th Award for Distinguished Technology in FY 2014 from the Atomic Energy Society of Japan (AESJ).