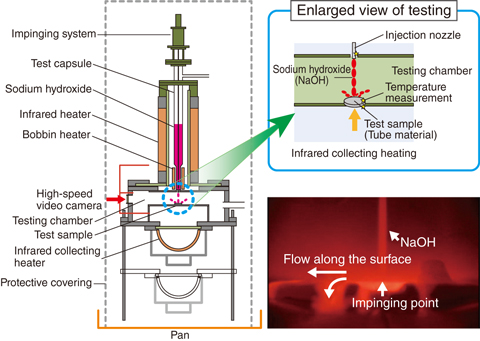
Fig.7-7 Experimental apparatus and situation
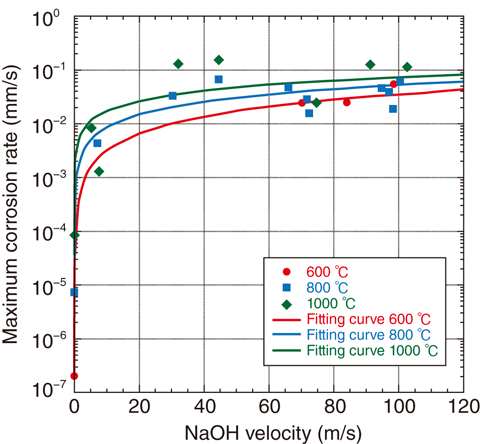
Fig.7-8 The corrosion rate in a high-temperature sodium hydroxide environment
A steam generator (SG) is a heat exchanger between sodium and water/water vapor in a sodium-cooled fast reactor (SFR). When the high-pressure water/water vapor leaks into sodium through a perforation breach of the tube, a high-temperature and highly caustic specific environment (reaction jet) may be formed. This reaction jet locally causes wear upon the neighboring tubes; this failure propagation may potentially reduce the structural integrity.
The wastage rate has thus far been evaluated on the basis of the data obtained in previous sodium-water-reaction experiments, which simulated the structure and operational conditions of existing SFR SGs. This approach is not a versatile option for design of future systems with high-temperature and high-pressure operation and is inadequate for optimization purposes. Therefore, a mechanistic sodium-water reaction-analysis system has been developed for the reasonable and socially-receptive design.
The authors proposed that wastage occurs as a combination of liquid droplet impingement erosion under the high-velocity reaction jet and corrosion induced by the sodium hydroxide (NaOH) and sodium monoxide (Na2O) flow mainly produced by the sodium-water chemical reaction. The experimental apparatus shown in the Fig.7-7 was developed to elucidate the separation effect of the factors influencing wastage behavior. This experimental apparatus primarily consists of a testing capsule to enclose the reagents such as NaOH and Na2O, an infrared heater to heat the testing capsule, a test sample manufactured of the same material as the candidate tube, an infrared-collecting heater to heat-test samples, and ancillary devices such as a data-acquisition system. The quartz-glass pipe was mounted between the infrared heater and the test capsule to sustain the argon-gas atmosphere in the experimental system. Experimental parameters include the temperatures of the reagent and the test sample (up to 1000 °C), the reagent-injection velocity (up to 100 m/s), and the reagent-mixture fraction. High-temperature liquid reagent impacted with the column at the predetermined position, as shown on the right side of Fig.7-7. The corrosion rate was measured by the reduced thickness of the test sample and the injection time.
Fig.7-8 indicates the maximum corrosion rate of the test sample in a NaOH environment. This rate tends to increase with the rise of reagent temperature, test-sample temperature, and reagent-impinging velocity. It was confirmed that the reagent-impinging velocity did not strongly affect the maximum-corrosion rate above approximately 30 m/s. This corrosion depression effect implies that the mass-transfer coefficients of iron and chromium, which are the reactants of the surface oxide, are much smaller within the material than those from the outer surface of the test sample to the outside fluid flow.
The results of this research will be utilized in the verification and validation of the mechanistic sodium-water reaction-analysis system.
The present study includes results obtained for the Ministry of Education, Culture, Sports, Science and Technology of Japan (MEXT).