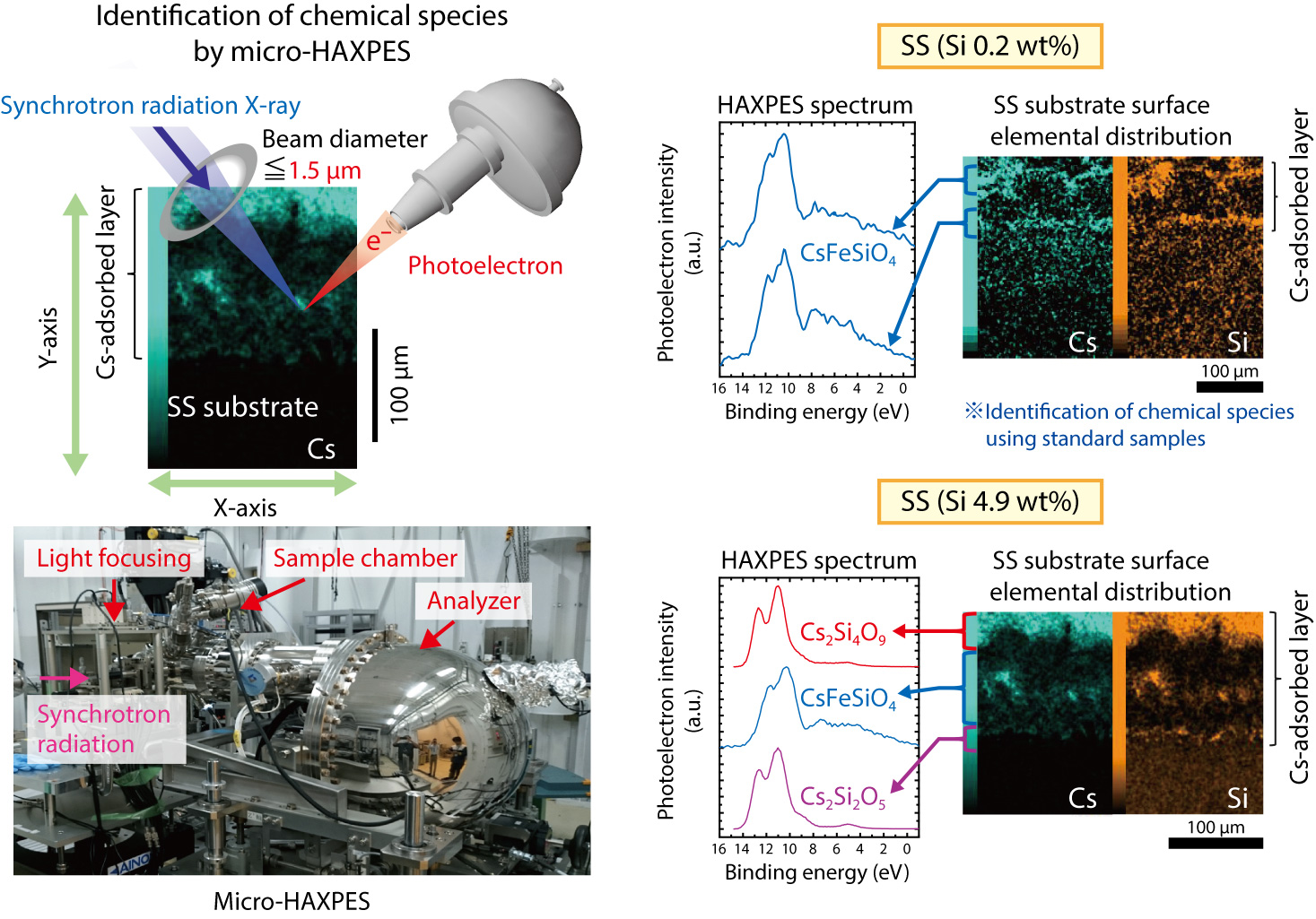
Fig.1-7 Result of identification of chemical species by micro-HAXPES for Cs-adsorbed stainless steel
At the TEPCO’s Fukushima Daiichi NPS, information on cesium (Cs) distributions within the nuclear reactors is necessary for managing the radiation exposures of workers, because Cs is a major source of γ ray. In particular, evaluation of Cs-chemisorption behavior, which is a reaction between Cs vapor and the stainless steel (SS) used in structural materials, is essential. Thus, such Cs-chemisorption amounts are estimated using severe-accident-analysis codes with Cs-chemisorption models. However, there is a large discrepancy among the analytical codes. One reason for this is that the chemical forms treated in their models differ.
Since Cs has various compounds with differing vapor pressures and water solubilities, the revaporization and transport behaviors of Cs from structural materials to gaseous and aqueous phases significantly depend upon which Cs compounds are formed. Therefore, clarification of the chemical forms of Cs adsorbed onto SS, which is widely used inside reactors, or clarification of reactions between SS and Cs vapor, is important for improving the estimation of Cs distributions within nuclear reactors. However, although Cs has been found to react with silicon (Si) impurities in SS, various Cs silicates have been reported and the major chemical form remains unclear, possibly due to the micron sizes of the reaction products.
Thus, we aim to develop an analytical technique to identify the chemical forms of micro-sized compounds by combining micro-hard-X-ray photoelectron spectroscopy (HAXPES) with scanning electron microscopy (SEM). Furthermore, since the chemical forms may depend upon the Si contents in SS, Cs chemisorption tests were performed using different Si-content samples. As a result, the compounds formed by chemical reaction between SS and Cs vapor were successfully identified, and their distributions were clarified (Fig.1-7). In particular, complicated chemical reactions were found to occur since not only iron (Fe)-containing Cs compounds (CsFeSiO4), but also Cs2Si2O5 and Cs2Si4O9, were formed when the Si content of SS increased.
In the future, we will try to elucidate the chemical properties of the identified Cs compounds such as vapor pressure and water solubility, and contribute to improving estimation of the Cs distribution within nuclear reactors by establishing a chemisorption model considering the chemical factors influencing the revaporization and transport behaviors of Cs.