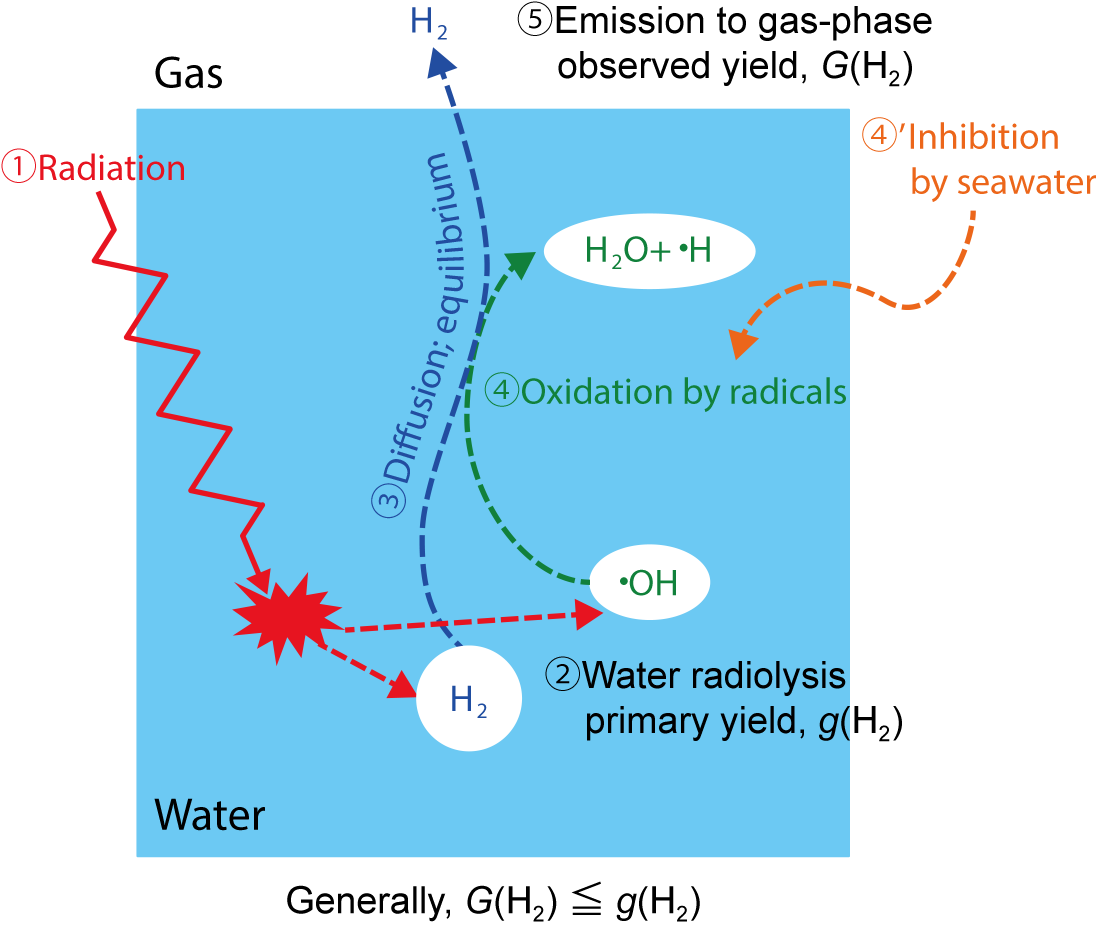
Fig.1-14 Diagram of radiolytic-H2 formation and reaction in water
Table 1-1 H2-generation factors relevant to the decommissioning of 1F
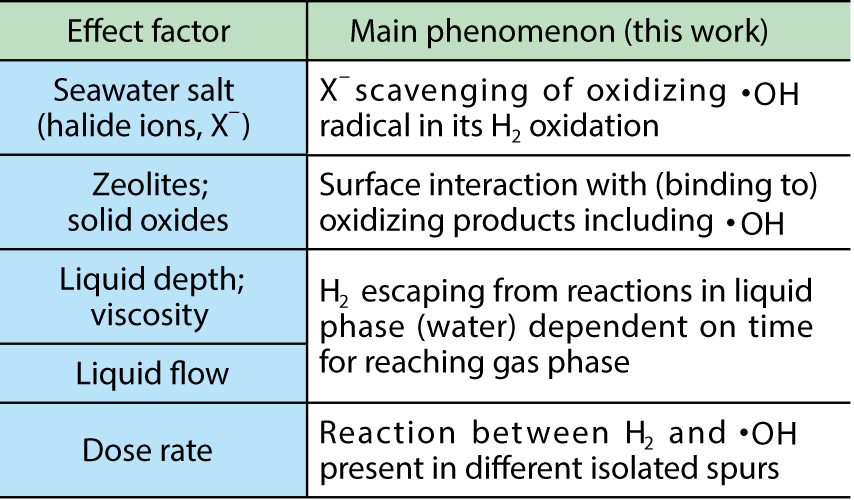
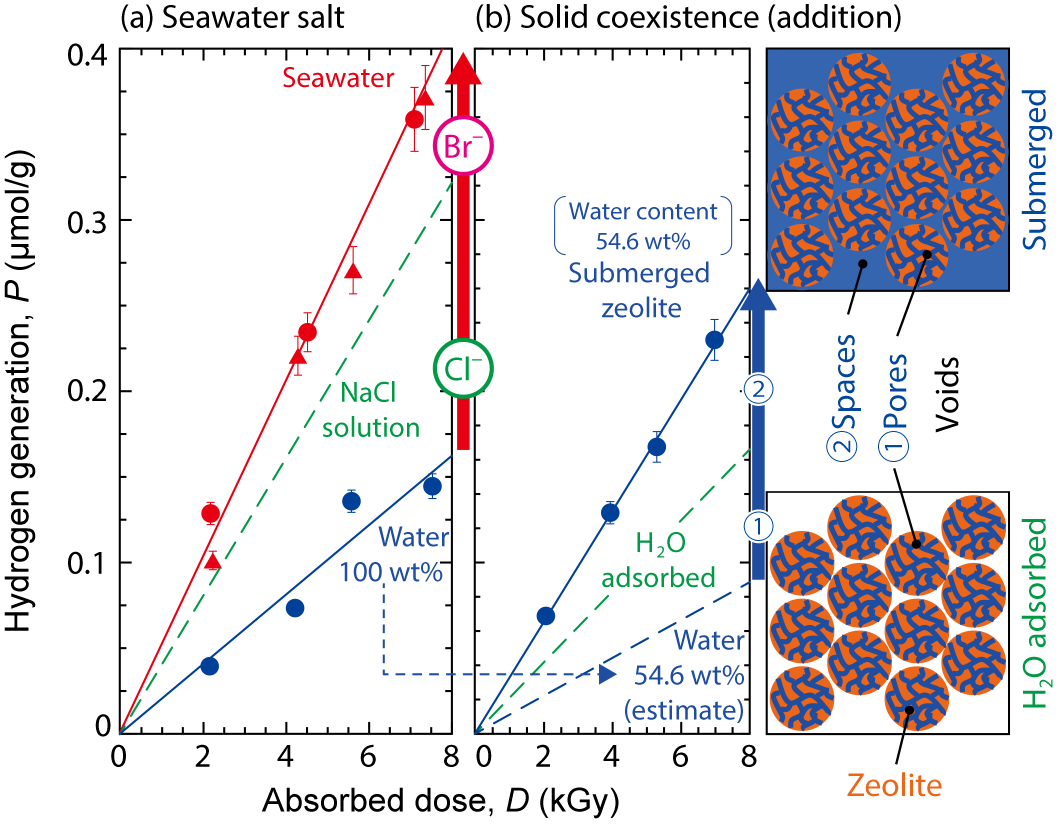
Fig.1-15 Effects of seawater salt (a) and solid coexistence (b) upon H2 generation (constant-height (1–cm) condition)
Following the accident at the TEPCO’s Fukushima Daiichi NPS (1F), we have focused upon water decontamination and secondary-waste storage, and performed R&D on cesium adsorption, hydrogen (H2) generation, corrosion of structural materials, and gas and heat flows. We then provided the data obtained as information indispensable for safety measures to TEPCO and 1F-related companies. Moreover, according to the medium- and long-term roadmap for decommissioning 1F, we have quickly complied with requests for studying and solving urgent problems and events in cooperation with TEPCO and the companies.
R&D upon H2 generation is related to the explosion of this gas just after the accident; it is considered to be the most dangerous phenomenon in the management of radioactive materials. Since water radiolysis in H2-generation sources is brought about by ionizing radiations emitted from radioactive materials, the temperature need not be higher than several hundred degrees centigrade; this process takes place continuously anywhere the materials for it exist. Thus, studies on water radiolysis have been performed at every step of the roadmap.
Unlike that derived from other sources, H2 formed from water decomposition by radiation is emitted into the air through numerous processes and reactions (Fig.1-14). H2 generation is complexly affected by many conditions and factors. Using practical materials under real conditions, several effects upon the generation have been investigated in detail and determined to be important for 1F decommissioning (Table 1-1).
Fig.1-15 shows representative experimental results. Seawater supplied at the accident works as a coolant as well as pure water (PW), while more H2 was generated in seawater than in PW (a). This was because chloride (Cl-) and bromide (Br-) ions in seawater suppress H2 consumption. The full amount of H2 generation in zeolites submerged by PW could not be explained only by their water content (b). This enhancement indicates an interaction between the radiolysis products of water and the solid surface of zeolite, giving useful information for improvement in conventional safety analysis of the generation without solids.
In these radiolysis studies, hydrogen peroxide (H2O2) and its decomposed product of oxygen (O2) are also studied.