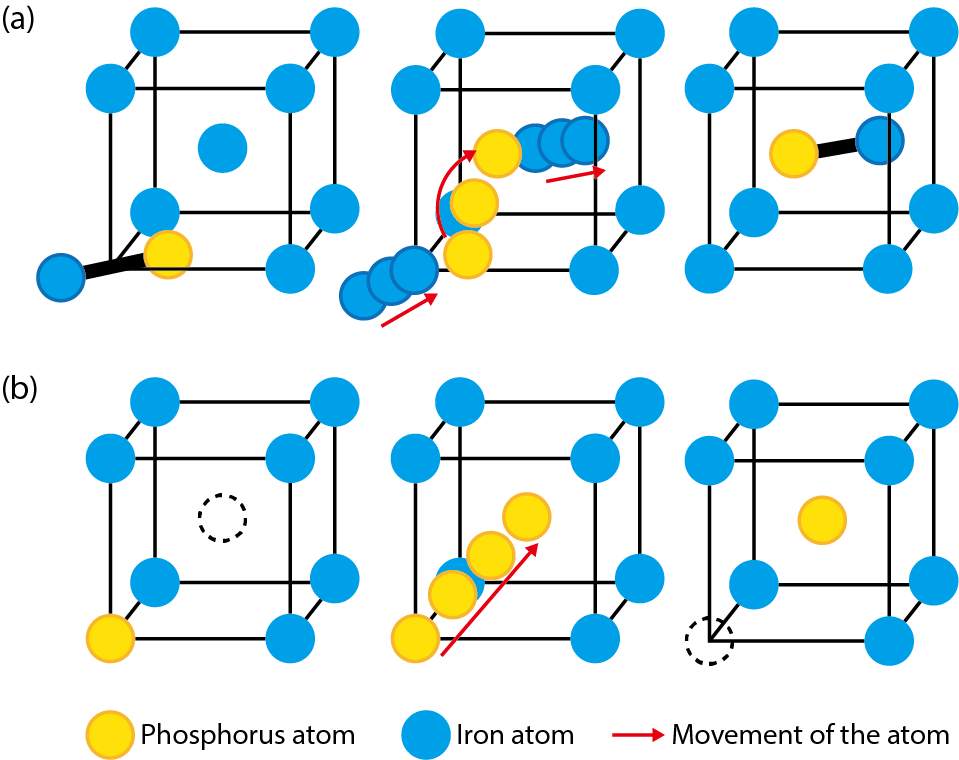
Fig.9-3 Movement mode of the phosphorus atom
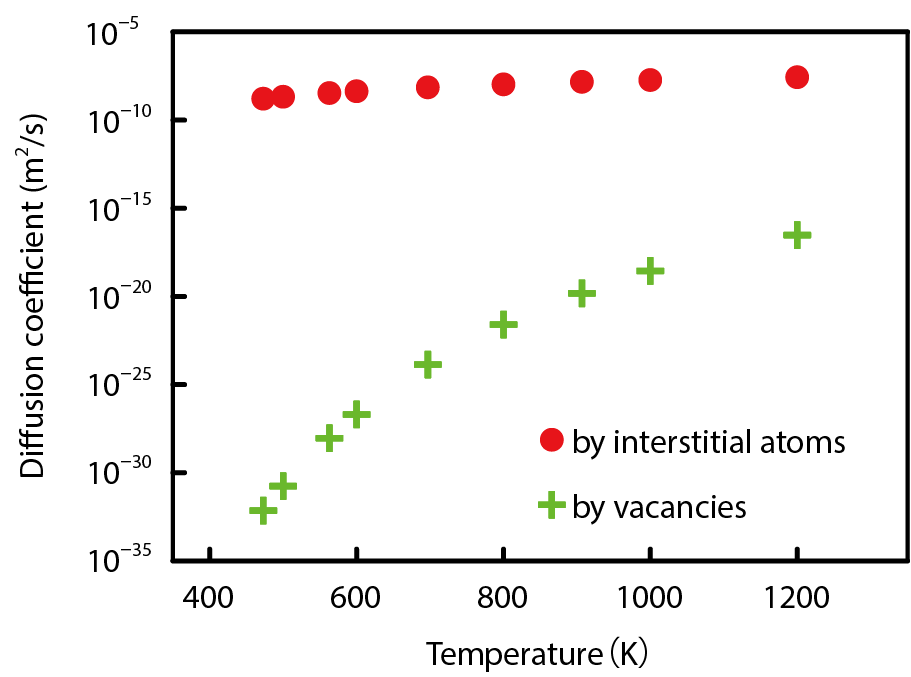
Fig.9-4 Temperature dependence of the diffusion coefficient evaluated by the simulation
Neutron irradiation at high temperature during reactor operation causes iron atoms in the stable positions of the crystal lattice in steels covering reactor cores to be repelled and move around. Along with this phenomenon, impurity atoms also start to gather at grain boundaries where the crystal lattice structure is in disorder, thereby weakening these boundaries. This phenomenon is called embrittlement, and it may eventually lead to cracking. Therefore, to safely operate nuclear reactors, it is important to estimate the amount of impurities at grain boundaries by evaluating how impurity atoms migrate and how fast they gather at these boundaries.
Phosphorus is one impurity that causes embrittlement at grain boundaries in steels. A phosphorus atom pairs with an interstitial iron atom moving via a gap between lattice nodes and can move by exchanging its partner to another atom (Fig.9-3(a)). On the other hand, the lattice node previously occupied by an expelled iron atom becomes a vacancy, and this vacancy moves when it is filled by an adjacent iron atom. When a vacancy moves to the node next to a phosphorus atom, this atom can move by jumping to the node of the vacancy (Fig.9-3(b)). These movements become more active as temperature rises. The moving mode of phosphorus atoms shown in Fig.9-3 can be found by consideration based on the energies of various configurations of atoms obtained from the first-principles calculation. However, one cannot know the diffusion speed from only the movement mode. Therefore, in this study, the movement of atoms was simulated by the kinetic Monte Carlo technique based on Fig.9-3, the time and distance of the movement of phosphorus atoms was measured, and the diffusion coefficient at various temperatures was calculated to evaluate the diffusion speed (Fig.9-4). Thus, it was confirmed that the movement by interstitial atoms is several orders of magnitude faster than that by vacancies. This result is useful for predicting the amount of phosphorus at grain boundaries under various conditions of temperature and irradiation as well as for evaluating the embrittlement of steels.
This method, which aims at evaluating macroscopic embrittlement based on atomic-level behavior that is difficult to measure experimentally, is called the multiscale modeling and is an effective computer-research method. In the future, we hope to contribute to the evaluation and prediction of nuclear-structural-material embrittlement by further improving this method.
The present study was partly supported by the Japan Society for the Promotion of Science (JSPS) KAKENHI Grant-in-Aid for Scientific Research (C) (No.15K06429).