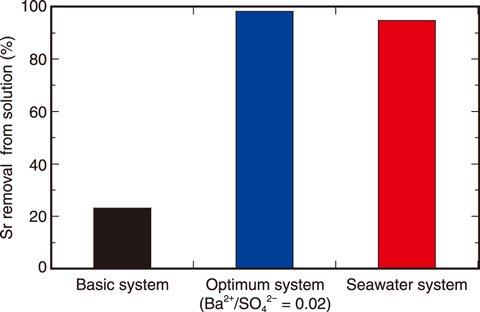
Fig.3-4 Strontium (Sr) removal by coprecipitation with barite
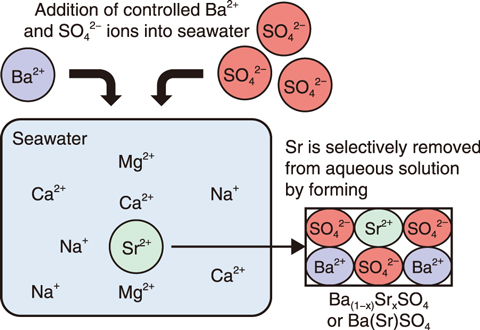
Fig.3-5 Experimental design for removing Sr from seawater by coprecipitation with barite
Strontium-90, which is a fission product of uranium and can be released to the environment during nuclear plant accidents, has a low adsorption capacity on clays and a high mobility in environments. There are few effective techniques for removing Sr from seawater because of the strong inhibition of Sr uptake by the high concentration of sodium and other competitive ions in seawater. In this work, barite (BaSO4) was used under various experimental conditions to develop techniques for the direct removal of Sr from seawater.
Barite is a common compound in many geological environments and easily precipitated from an aqueous mixture of Na2SO4 and BaCl2. Several trace elements, including Sr, can be incorporated into the crystal lattice of barite during a coprecipitation process. The effects of pH, ionic strength (IS), coexistent Ca2+ and Mg2+ ion, and [Ba2+]/[SO42-] ratio in the initial solution were investigated for determining the optimum condition for Sr removal by using barite. The resulting removal efficiency for three of the studied systems is shown in Fig.3-4. The main parameter found to affect the removal efficiency was the [Ba2+]/[SO42-] ratio in the initial aqueous solution; Sr was completely removed from the aqueous solution when the [Ba2+]/[SO42-] ratio is low. A high removal efficiency of 90% was also achieved in seawater (=1 mg/L), despite its high saline concentration, due to the negligible effect of IS and competitive ions (Na+, Ca2+, and Mg2+). Thus, compared to previous techniques, barite was shown to present a reliable material for the removal of Sr from aqueous solutions, including seawater, after optimization of the [Ba2+]/[SO42-] ratio and other parameters (Fig.3-5).
This method can be applied to other radioactive anions present in high-level waste at Fukushima, such as selenium-79 and iodine-129, for safety assessments of long-term geological disposal.