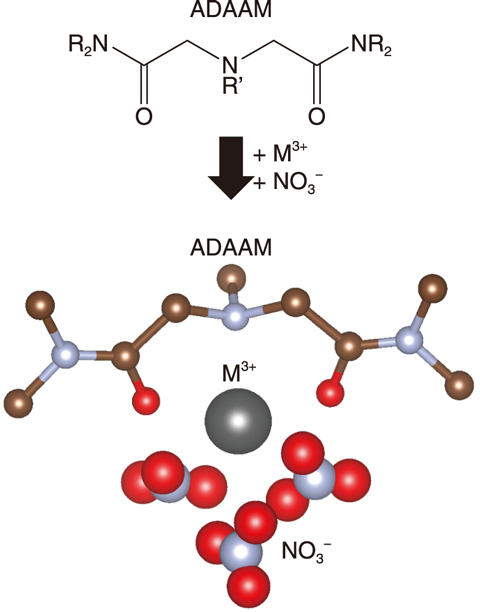
Fig.4-7 Structure of complex Am/Cm ion and an ADAAM separation ligand
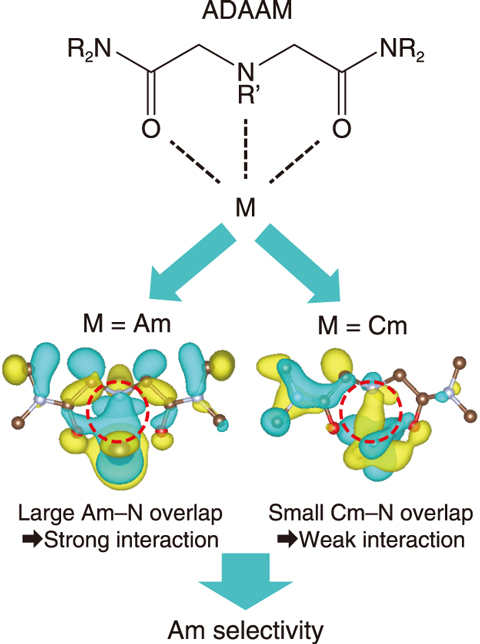
Fig.4-8 Interaction between Am/Cm ions and a molecular of ADAAM ligand
To reduce the toxicity of high-level radioactive liquid waste (HLLW), the partitioning of minor actinides (MAs), which have extremely long half-lives, and the transmutation of short-life nuclides, partitioning and transmutation, has been developed. Although americium (Am) and curium (Cm) should be separated during partitioning due to the heat generating property of Cm, their similar chemical properties have made separation difficult. Therefore, We developed a process to selectively separate Am from Cm using alkyldiamideamine (ADAAM) as a separation ligand. Furthermore, the mechanism by which ADAAM shows higher selectivity of Am over Cm was clarified using a quantum chemical calculation.
Am and Cm exist as trivalent ions, Am3+ and Cm3+, respectively, and form complexes with separation materials. The complex consisting of the metal ion (M3+), nitrate ion (NO3-), and ADAAM was modeled as [M(ADAAM)(NO3)3] (Fig.4-7), by referring to the result of the previous separation experiment (*1, *2). The Gibbs energy difference on the formation of each complex was then calculated to indicate that the Am complex is more stable than the Cm complex. The calculated value of the separation factor of Am from Cm was 6.2 and reproduced the experimental value, 5.5.
The coordination bonds between the metal ion and ADAAM were then examined with the aim of understanding the higher observed Am selectivity. ADAAM forms bonds with the metal ion using one amine nitrogen atom and two carbonyl oxygen atoms. Calculated result of coordination geometry revealed that the bond lengths between the metal ion and nitrogen atom of the ADAAM were 2.91 and 2.94 Å for the Am and Cm complexes, respectively. Furthermore, the covalent interactions of the coordination bonds were then analyzed by calculating the overlap of electron orbitals between the metal ion and nitrogen atom. The strength of the covalent interaction between atoms represents the degree of the overlap between electron orbitals. The overlap of the Am-N bond was larger than that of the Cm-N bond (Fig.4-8), thus indicating that the difference in the bonding strength between the metal ion and the ADAAM is a key to understand the Am/Cm selectivity. This work and future work on the selectivity in the separation of MAs is expected to contribute to the development of materials used for separating metal ions.
This work was supported by the Japan Society for the Promotion of Science (JSPS) KAKENHI Grant-in-Aid for Young Scientists (B) (No.17K14915).