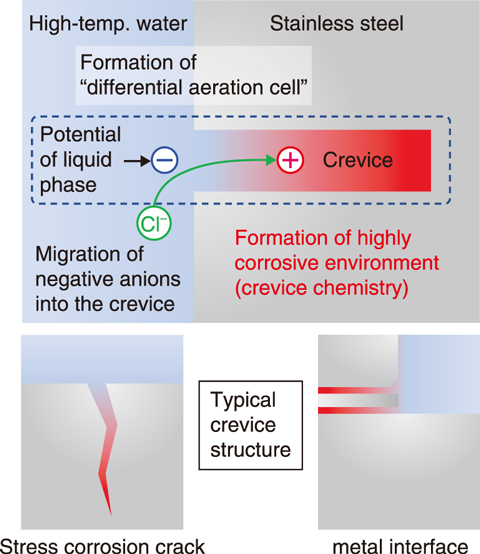
Fig.4-4 Schematic representation of chemical condition within a crevice
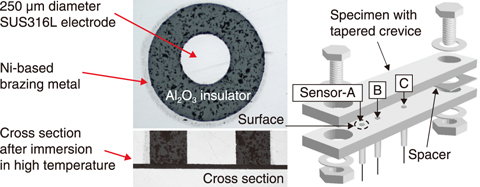
Fig.4-5 Developed sensor systems to measure the crevice chemistry
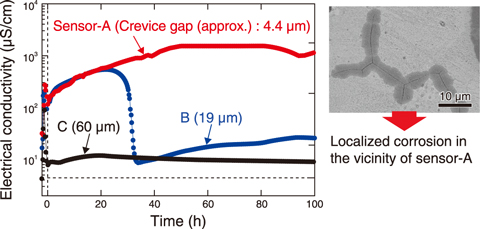
Fig.4-6 Electrical conductivity of the solution within the crevice in 288 ℃ pure water with dissolved oxygen concentration of 32 ppm
Stainless steels are susceptible to stress corrosion cracking (SCC) in high-temperature and high-pressure water represented by boiling water reactor (BWR). SCC of stainless steels have been recognized as a major corrosion related problems of ensuring safety of nuclear power plants. The chemical conditions within a crevice including stress corrosion crack may become more corrosive than those of the bulk coolant (Fig.4-4), and this plays an important role with regard to crack propagation. The chemical conditions within a crevice often referred to as crevice chemistry have been widely studied, however, analysis of the crevice chemistry is still challenging because crevice with micro-meter gap under high temperature and high pressure is experimentally difficult to access.
A sensor system was thus developed to conduct measurement of the crevice chemistry (Fig.4-5). The system is characterized by high durability in the harsh environment, uses small electrodes to allow for measurement in very tight crevices, and in-situ analysis of the chemistry by electrochemical method. The sensors were installed at different positions within a tapering crevice of Type-316L stainless steel and measurement of the local electrical conductivity of the crevice solution beneath the sensor was carried out.
Fig.4-6 shows the time variation of electrical conductivity of the crevice solution at different position in 288 ℃ and 8 MPa high purity water. The results indicated that smaller crevices led to higher conductivity; sensor-A, with a gap of 4.4 μm had conductivity values over 100 times greater than those at sensor-C, with a gap of 59.3 μm, and sensor-B with an intermediate gap detected high conductivity comparable to sensor-A up to 20 h, but thereafter, it dropped rapidly. Localized corrosion occurred in the vicinity of the sensor-A. Thermodynamic consideration indicated that the high conductivity values corresponds to the acidification of the solution. Thus, corrosive environments were formed in crevice with small gaps and caused the localized corrosion.