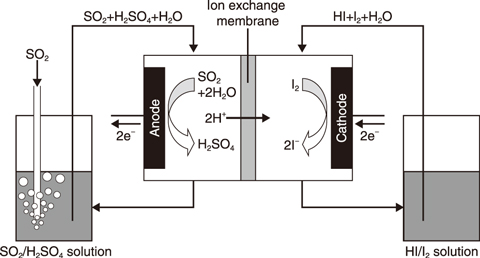
Fig.6-15 Schematic of Bunsen reaction using ion-exchange membrane
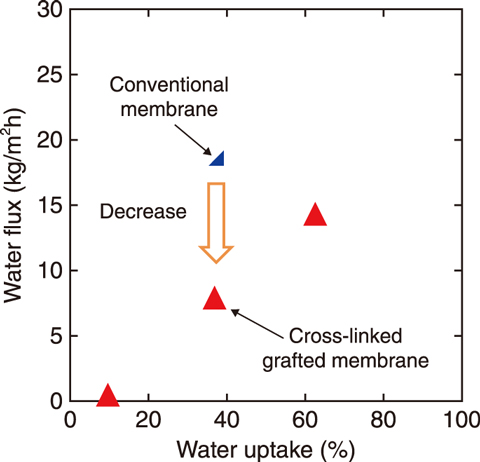
Fig.6-16 Relationship between water permeate flux and water content

Fig.6-17 Outlook of (a) conventional and (b) cross-linked grafted membrane after membrane Bunsen reaction tests
In an effort to valorize the heat of high-temperature gas-cooled reactors, the thermochemical iodine-sulfur (IS) water-splitting hydrogen (H2) production process has been investigated. The IS process is a promising H2 production method using nuclear and renewable energy sources and is composed of sulfur and iodine chemical reactions. The Bunsen reaction, a key reaction of the IS process, produces sulfuric and hydriodic acids by mixing iodine (I2) and sulfur dioxide (SO2) with water. Liquid-liquid (LL) separation occurs by adding excess I2 to the two acids. However, this excess I2 decreases process efficiency and increases cost.
One alternative Bunsen reaction method investigated involves using an electrochemical cell equipped with a proton-selective ion-exchange membrane (Fig.6-15). No LL separation is needed, as the two acids can be separately produced in a cell partitioned into two channels by the ion-exchange membrane.
An ion-exchange membrane suitable for this purpose should have a high conductivity and low water permeability. Enhancing conductivity requires higher water content in the membrane however, this causes higher water permeation. A cross-linked ion-exchange membrane was therefore prepared by a radiation grafting technique developed by National Institutes for Quantum and Radiological Science and Technology (QST). The cross-link, in which grafted chains connect, enables the membrane to retain water among the chains.
The relationships between water permeate flux and water content of the trial membranes were then measured (Fig.6-16); a lower flux was found of the ion-exchange membrane than of a conventional membrane having the same water content. Membrane Bunsen reaction tests were carried out using an electrochemical cell introducing the tested membranes, revealing a lesser degree of white turbidness of the cross-linked grafted membrane (Fig.6-17(b)) than that of a conventional membrane (Fig.6-17(a)). Thus, a cross-linked grafted membrane can reduce water permeation and the white turbidness due to sulfur formation associated with SO2 transportation accompanied with water, indicating that a cross-linked grafted membrane can be adapted to the membrane Bunsen reaction. Future work will involve the development of an ion-exchange membrane to improve performance by using this membrane preparation technology.
This study was partly supported by Council for Science, Technology and Innovation (CSTI), Cross-ministerial Strategic Innovation Promotion Program (SIP).
<Previous: 6-6 | Next: 7 Research and Development of Fast Reactors>