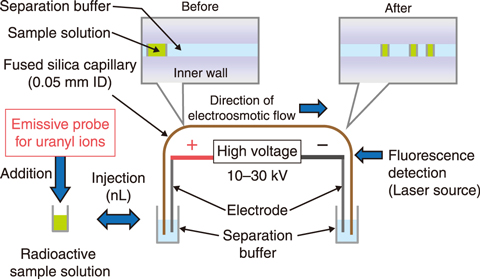
Fig.8-4 A simple analytical method with capillary electrophoresis
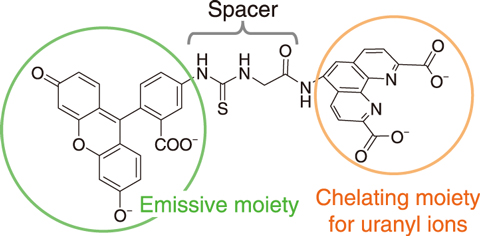
Fig.8-5 Emissive probe L for uranyl ions
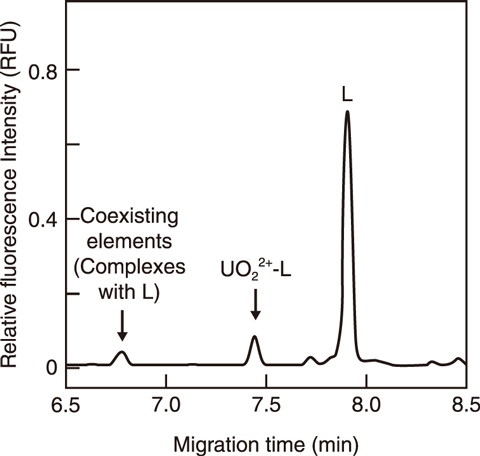
Fig.8-6 Typical electropherogram of a uranyl complex with probe L
It is necessary to evaluate the radioactivity concentrations of radionuclides in samples collected from radioactive wastes to ensure their safe disposal. To reduce exposure of operators to radiation during analytical procedure, a simple analytical method for uranyl ions was developed using capillary electrophoresis (CE) (Fig.8-4). Although CE has been employed to efficiently and easily separate ionic species due to their difference in electrophoretic speed caused by ionic charge and size differences in a small-diameter capillary tube, the sensitivity of prior methods using absorbance detection was too low for practical usage in radioactive waste samples.
This study therefore focuses on a capillary electrophoresis-laser induced fluorescent detection method (CE-LIF) to improve the sensitivity by a factor of approximately one million, from the level of ppm to that of ppt. A new emissive probe suitable for CE-LIF was designed to detect uranyl ions, UO22+. The emissive probe was composed of an emissive moiety for improved sensitivity, a chelating moiety for connecting with uranyl ions and a spacer between them. A chelating moiety is required for stability not to dissociate from uranyl ions during migration. Emissive probes synthesized with multidentate chelating moiety were applied to CE-LIF and to investigate the stability of the chelating moiety based on experimental studies and molecular orbital calculations. As a result, a tetradentate probe (Fig.8-5) achieved a successful separation and detection of uranyl ions (Fig.8-6). A detection limit of 0.7 ppt was obtained for uranyl ions. Therefore, the developed fluorescent detection method was one million times more sensitive conventional UV absorbance detection methods. The established CE-LIF method was then applied to radioactive liquid samples obtained from a JAEA radioactive waste treatment facility; the ability to separate and detect uranyl ions in coexisting elements was thus demonstrated. This method, requiring only a small sample volume and analytical time within ten minutes, can be used to analyze radioactive samples to reduce the radiation exposure to operators.
This research was supported by the Japan Society for the Promotion of Science (JSPS) KAKENHI Grant-in-Aid for Young Scientists (B) (No.16K17926), “Development of highly sensitive and rapid analytical method for ultratrace actinide ions in radioactive samples by capillary electrophoresis”.
<Previous: 8 Research and Development on Fuel Reprocessing, Decommissioning, and Radioactive Waste Management | Next: 8-2>