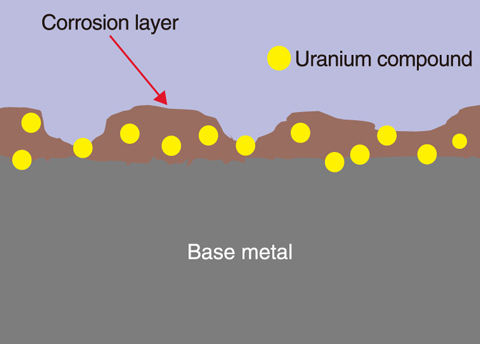
Fig.8-7 Concept of a metal surface contaminated by a uranium compound
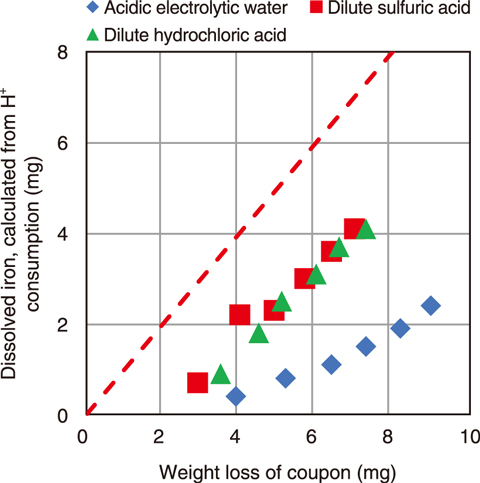
Fig.8-8 Dissolution characteristics of carbon steel coupons with a corrosion layer
To reduce costs and environmental burdens, decontamination technologies have been under development to reduce uranium waste generation. As the metal waste generated from dismantling uranium handling facilities can be contaminated with uranium, they must be decontaminated to the clearance level and then reused or disposed of as non-radioactive waste.
As metal waste encompasses a variety of materials, shapes, and surface conditions, a wet decontamination method using dilute sulfuric acid is generally applied. However, this process generates excessive secondary waste and many metals, such as carbon steel, is excessively dissolved.
A corrosion layer is easily generated on carbon steel, the main material of centrifuges used for uranium enrichment, when exposed to atmospheric water during dismantling. As the corrosion layer is formed on surface by uptake of oxygen from the atmosphere, most of the uranium on the surface has moved to the corrosion layer as shown in Fig.8-7, but a small quantity of uranium diffuses into the base metal.
Removing the corrosion layer with the conventional decontamination method is time-consuming. The added exposure time of the metal to the acid used causes secondary waste containing dissolved base metal.
The applicability of acidic electrolytic water (AEW) as a decontamination solution to dissolve uranium and minimize the dissolution of the base metal was examined. The AEW used contained approximately 100 ppm of hydrochloric and hypochlorous acid, resulting in a pH of 2.5-2.8 and an oxidation–reduction potential above 1100 mV. The following results were obtained.
(1) Weigh loss to the amount of iron dissolution for AEW was the largest in simulant coupons test. Thus, the corrosion layer was removed more effectively than when using dilute sulfuric acid (Fig.8-8).
(2) The target surface decontamination density (0.04 Bq/cm2) was achieved in approximately half the time when AEW combined with ultrasonic cleaning was used rather than when other acids were used.
(3) Approximately 20% less secondary waste was generated when AEW, due to the difference of the amount of iron components in the solutions.
This study was conducted in collaboration with Japan Nuclear Fuel Limited. We will continue to work with this company on the practical use of metal decontamination technology.