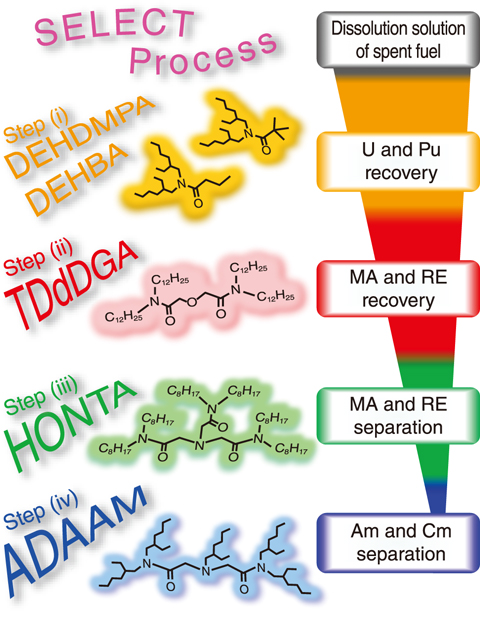
Fig.4-13 Schematic of SELECT process and extractants used
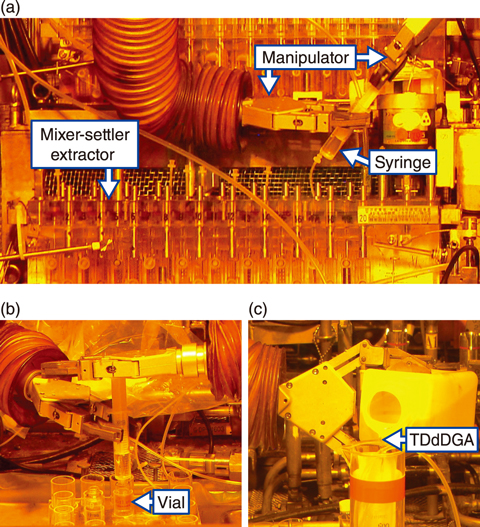
Fig.4-14 Recovery of MA and RE performed in a concrete cell
To reduce the radiotoxicity and volume of high-level radioactive waste, separation and transmutation technologies by which some radioactive isotopes are transmuted using an accelerator-driven system after separation from a dissolution solution of spent nuclear fuel or high-level liquid waste have been under development. The separation of americium (Am) and curium (Cm), designated here as minor actinides (MAs), is necessary to achieve this goal, as MAs consist of isotopes with high radioactive toxicity or a high heat-generation rate.
Many of the solvent separation processes developed to date use extractants consisting of phosphorus and sulfur; however, such extractants are difficult to decompose into gases by incineration, and thus can increase secondary waste. To overcome this challenge, a separation process using extractants comprising carbon, hydrogen, oxygen, and nitrogen was developed; this process, entitled SELECT (Solvent Extraction from Liquid waste using Extractants of CHON-type for Transmutation), is summarized schematically in Fig.4-13, including the extractants used in each step. Since the dissolution solution of spent nuclear fuels and high-level radioactive liquid waste consist of various elements, single-step separation is difficult. The SELECT process, therefore, consists of four steps.
In Step (i), uranium (U) and plutonium (Pu) are recovered from the dissolution solution of spent nuclear fuel; they can be reused as nuclear material. The MAs and rare earth elements (REs) are then recovered from other fission products in Step (ii), and thereafter MA and RE are separated from each other in Step (iii). Finally, Am and Cm are then separated in Step (iv).
The successful demonstration of Step (i) has been performed via continuous counter-current experiments, in which monoamides such as DEHBA and DEHDMPA were used as extractants. A continuous counter-current experiment was thus designed and carried out to validate Step (ii) using a high-level liquid waste obtained from the demonstration of Step (i) (Fig.4-14). Although TODGA has been commonly employed as an extractant to recover MA and RE from high-level liquid waste, TDdDGA was used here, due to its demonstrated higher extraction capacity. Due to the high radioactivity of the experimental solution, the experimental apparatuses were installed in a concrete cell and were operated by manipulators.
The resulting recoveries of the MAs and REs were >98% and 86.9–99.9%, respectively, thus demonstrating the validity of Step (ii). For further development of SELECT process, studies on Step (iii) and Step (iv) using HONTA and ADAAM as extractants, respectively, are ongoing.
(Yasutoshi Ban)