
Fig.1-6 (a) Schematic image and appearance of simulated MCCI product, (b) optical micrograph of a cross section of the simulated MCCI product, (c) SEM image of the upper region in the once-melted zone, and (d) SEM image of the bottom region in the once-melted zone
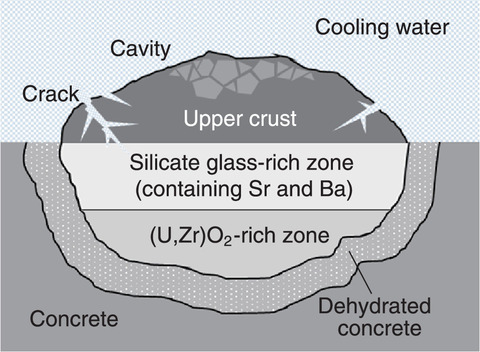
Fig.1-7 Image of layered structure of the MCCI products
In the severe accident at TEPCO's Fukushima Daiichi NPS (1F), the molten corium consisting of nuclear fuels and structural materials was predicted to spread over the concrete floor of the primary container vessel, resulting in molten core-concrete interaction (MCCI). The MCCI products continue to be exposed to cooling water. Under such conditions, the water-soluble radionuclides may be gradually dissolved from the MCCI products, and then, some of these radionuclides will likely migrate via water. Knowing the structure of MCCI products and the distribution of such radionuclides in MCCI products is important to understanding the diffusion behavior within 1F. In this study, we focused on Sr and Ba, which have high radioactivity. A reaction test between simulated corium (containing nonradioactive Sr and Ba) and concrete was performed and information on the structure of MCCI products and the distribution of Sr and Ba was experimentally obtained.
Fig.1-6 (a) shows a schematic illustration and appearance of the simulated MCCI product. The powder mixture of simulated core materials (U0.5Zr0.5O2, ZrH2, stainless steel (SS), B4C, Mo-Ru-Rh-Pd, BaCO3, and SrCO3) was pressed into a tablet and placed on a basaltic concrete piece. Light from a Xe lamp (3 kW maximum) was directed onto the tablet surface using a light-concentrating heating device to form a simulated MCCI product under a vertical temperature gradient.
The solidified sample was cut in a vertical direction and Fig.1-6 (b) shows the optical micrograph of the cross section. The corium and concrete were interacted as liquid phases and solidified in the upper gray region (once-melted zone). Below the once-melted zone, concrete did not melt but thermally dehydrated. Figs.1-6 (c) and (d) show SEM (scanning electron microscope) images of the upper region and bottom region of the once-melted zone, respectively. The main phases are dark silicate glass and bright (U,Zr)O2 particles. Sr and Ba were detected in the silicate glass (approximately 3at%-Sr and 2at%-Ba), whereas they were not detected in (U,Zr)O2. Thermodynamic analysis reveals that Sr and Ba tend to dissolve in silicate glass, rather than in (U,Zr)O2.
(U,Zr)O2 precipitated as fine particles (2-3 µm) in the upper region, while it precipitated as bigger particles (10-20 µm) in the bottom region. In addition, the content of (U,Zr)O2 is much higher in the bottom region. During the heating tests, sedimentation of (U,Zr)O2 particles may have occurred in the liquid silicate glass because of its higher density.
An image of the MCCI product layered structure is drawn in Fig.1-7. According to the 1F accident scenario, the crust might have formed on the top surface with cracks and cavities. The water possibly contacted the silicate glass-rich zone, where Sr and Ba are likely to be concentrated. Future research will include the leaching behavior of Sr and Ba from silicate glass. These data will contribute to the diffusion behavior of radionuclides within 1F.
(Ayako Sudo)