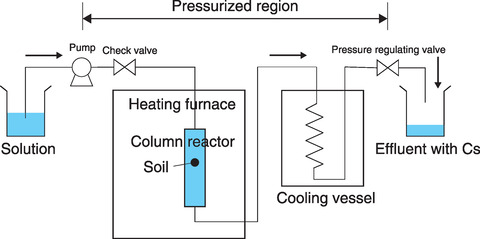
Fig.1-10 Hydrothermal treatment device to remove Cs from soil
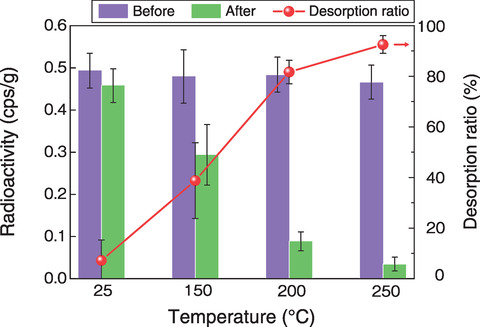
Fig.1-11 Removal of radioactive Cs from actual contaminated soil
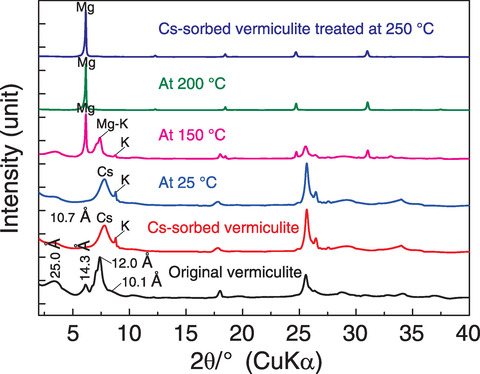
Fig.1-12 Ion exchange between Mg and Cs on vermiculite in subcritical conditions
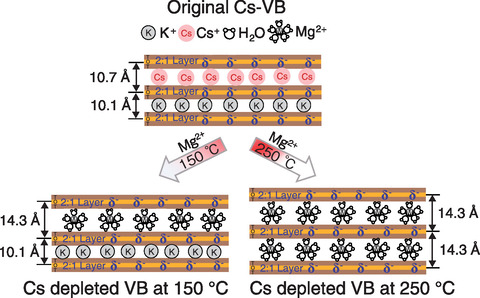
Fig.1-13 Reaction model of Mg exchange with fixed Cs in soil particles of vermiculitized biotite (VB)
The soil surrounding TEPCO's Fukushima Daiichi NPS had been contaminated with radionuclides that were released during the accident, including 137Cs. Volume reduction techniques of contaminated soil are needed for its storage and management until it can be feasibly disposed of in the future. As radioactive Cs in the soil might be selectively and strongly adsorbed onto the clay minerals of the local soil, sieving can be used to roughly separate the minerals from the soil and thus provide Cs enrichment. Nevertheless, novel chemical treatment techniques capable of removing Cs from clay are indispensable for effective soil decontamination.
Generally, Cs trapped by soil particles is fixed in the clay interlayer. Cation-exchange methods using highly concentrated salt solutions at temperatures above the boiling point of water can be used to remove this strongly bound Cs. However, Cs can be resorbed during cooling, making decontamination difficult and requiring that the soil particles are treated with leaching solutions and then separate them in subcritical and pressurized conditions (i.e., hydrothermal conditions). Removing Cs from soil requires the primary contact of soil particles with an aqueous solution under pressure and their fast separation from solution containing Cs.
Therefore, we conducted Cs removal tests via the continuous reaction of solid and liquid under high temperature and pressure. A column system was designed for Cs desorption in which a reactor was loaded with soil particles that were then leached at a constant temperature by continuous circulation of an aqueous solution, as shown in Fig.1-10. An Mg solution shown to be effective for Cs leaching in previous studies was used to remove Cs from contaminated soil. After treatment at ≥200 ℃, the soil radioactivity was greatly decreased, and the Cs removal ratio was >80%, as summarized in Fig.1-11. This Cs removal process by Mg exchange in Cs-sorbed vermiculite was then explored by XRD. As shown in Fig.1-12, no Mg-interlayer peak was present in the samples at 25 ℃; however, above 150 ℃, peaks representing the Mg- and K-interlayers were present in the treated samples, whereas the peak representing the Cs-interlayer (10.7 Å (=10-10 m)) was not observed. Further, the proposed method using Mg effectively removed Cs trapped in the layered vermiculite (i.e., achieved a higher removal ratio) at treatment temperatures ≥200 ℃ (Fig.1-13).
Overall, the proposed method presents an effective and feasible Cs removal chemical treatment for soil decontamination.
This work includes partial results from joint research with the Tokyo Institute of Technology titled “Research on radioactive material recovery from radioactive waste by subcritical water washing and solid-phase extraction”.
(Xiangbiao Yin, Yoshikazu Koma)