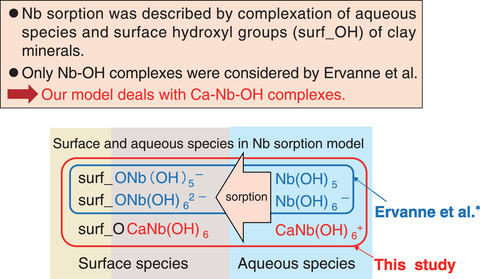
Fig.2-17 Conceptual drawing of Nb sorption model
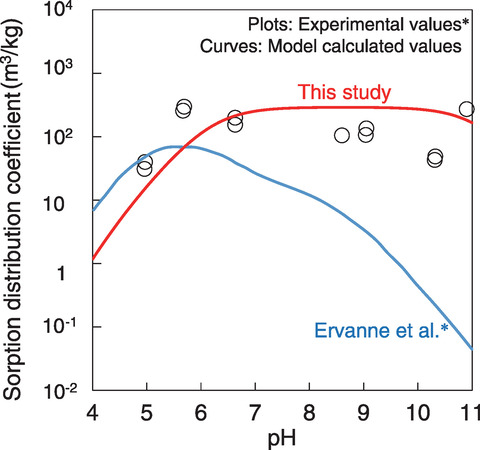
Fig.2-18 Model results for Nb sorption onto illite in 0.1 M Ca(ClO4)2 solution
Radioactive waste from activated reactor core internals is classified as relatively higher radioactive waste, which is a category of low-level waste. As these wastes contain long-lived radionuclides and must be kept away from the biosphere long-term, it is planned to be disposed of underground in Japan. Safety assessments of radioactive waste disposal must analyze the capacity of radionuclides leached from waste forms to migrate through the bedrock to the biosphere, where humans can be exposed to the waste. The migration of radionuclides is expected to be slowed by sorption onto rock-forming minerals. This process is affected by coexisting ions in groundwater, the compositions which may change according to the long-term disposal environment. Therefore, a reliable model must be developed to estimate the sorption properties of radionuclides onto rock-forming minerals.
Niobium-94 (94Nb) is a long-lived radionuclide (half-life: 20300 y) contained in the wastes of reactor core internals and important in the safety assessment of waste disposal because of its contribution to radiation doses. However, few researchers have examined the sorption of Nb onto minerals. Ervanne et al.* examined the effect of sodium (Na) and calcium (Ca) on Nb sorption onto illite, which is a type of rock-forming clay mineral with a high specific surface area (i.e., high sorption capacity). In the presence of Na, the experimental sorption data were reproduced by their proposed model, which assumed aqueous and surface species of Nb hydroxide complexes (Nbx(OH)y5x-y). However, their model did not agree with the data obtained in the presence of Ca.
Therefore, we developed a Nb sorption model to resolve this divergence by assuming the existence of Nb complexes with OH- and Ca2+ (Ca-Nb-OH complex), which had not been considered before. In order to confirm the formation of aqueous Ca-Nb-OH complexes, solubility experiments of Nb were performed under various Ca concentrations and pH. The experimental results showed that CaNb(OH)6+ was formed as the dominant aqueous species, in addition to Nb(OH)6- and Nb(OH)72-. Additionally, Ca-Nb-OH complexes containing surface OH- groups (surf_OH) were assumed to be formed and follow the sorption reaction: surf_O- + CaNb(OH)6+ = surf_OCaNb(OH)6. In this reaction, we excluded H+ because no pH dependence was observed in the sorption data. Therefore, CaNb(OH)6+ and surf_OCaNb(OH)6 were added to each species in the developed model (Fig.2-17). The Nb sorption onto illite in a 0.1 M Ca(ClO4)2 solution was then calculated using the geochemical calculation code (PHREEQC) with thermodynamic CaNb(OH)6+ data determined from the solubility experiments. The model results represented trends in the experimental data obtained by Ervanne et al.* that could not be explained by the previous model, as shown in Fig.2-18.
The results of this study will be used as a scientific basis for highly reliable evaluation of the sorption property of 94Nb to rock in the future disposal of the wastes from reactor core internals.
(Saki Ohira)
* Ervanne, H. et al., Modelling of Niobium Sorption on Clay Minerals in Sodium and Calcium Perchlorate Solutions, Radiochimica Acta, vol.102, issue 9, 2014, p.839-847.