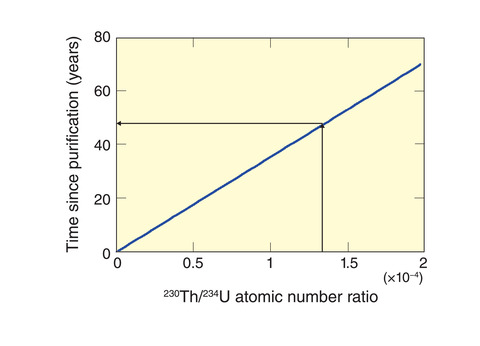
Fig.2-19 Relationship between the number of years since the purification period and the 230Th/234U atomic number ratio

Fig.2-20 Scanning electron microscope image of a single uranium particle
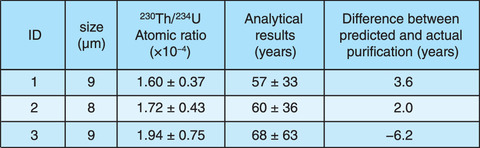
Table 2-2 Calculated time since purification using a single uranium particle
The International Atomic Energy Agency (IAEA) conducts safeguards to ensure that nuclear materials, such as uranium, are only used for peaceful (i.e., energy-related) purposes. One such activity comprises environmental sample analysis to detect undeclared nuclear activities by analyzing the composition and quantity of ultra-trace amounts of nuclear materials adhering to the special cloths that IAEA inspectors use to wipe down walls and other surfaces inside and outside nuclear facilities. The samples collected are analyzed by the IAEA Network Analytical Laboratories, of which JAEA is a part. The year of uranium purification can provide evidence of the existence of an undeclared nuclear development program; thus, researchers have aimed to develop analytical methods to estimate the year of uranium purification.
The uranium isotope 234U undergoes radioactive decay to thorium-230 (230Th) with a half-life of approximately 250000 years. As shown in Fig.2-19, the ratio of 230Th/234U atoms increases with time from purification; by analyzing this ratio, the time elapsed since uranium purification can be estimated.
However, the uranium particles captured on samples collected by IAEA inspectors are very small, weighing only nano-grams (10-9 g). In ordinary laboratories, accurate results cannot be obtained with such a small sample due to the influence of natural uranium in environmental dust. Therefore, a 230Th/234U atomic number ratio analysis technique using chemical separation was developed for use on a single uranium particle at the Clean Laboratory for Environmental Analysis and Research. The isotope dilution method was applied by adding a standard to the sample to obtain accurate atomic number ratios. A single standard with a known 229Th/233U atomic number ratio was prepared to ensure accurate values could be obtained without strictly controlling the amount added to the sample or the weight of the standard material. An image of a sample particle analyzed is shown in Fig.2-20. This particle was prepared from a reference material with a known date of purification. After the uranium particles were dissolved, standards were added, and chemical separation pretreatment was performed. The 230Th/229Th and 234U/233U ratios were then analyzed by inductively coupled plasma mass spectrometry (ICP-MS), and the 230Th/234U atomic number ratio was calculated to estimate the years since purification.
The obtained results are shown in Table 2-2. For particles with a diameter of 8 to 9 µm, the actual period of purification (61 years) was estimated with an error of between -6.2 and 3.6 years. Therefore, the proposed technique can accurately estimate the year of uranium purification from a single uranium particle, as required by the IAEA. Future efforts will include the use of ICP-MS equipped with high-sensitivity multiple detectors. The ultra-trace amounts of 230Th in uranium particles in the range of femto-grams (10-15 g) can be then determined with more accurately to estimate the year of purification. By applying this technology to the analysis requested by the IAEA, we hope to contribute to international safety and cooperation.
A part of this study was conducted in FY2020, sponsored by the Nuclear Regulation Authority (NRA), Japan.
(Daisuke Suzuki)