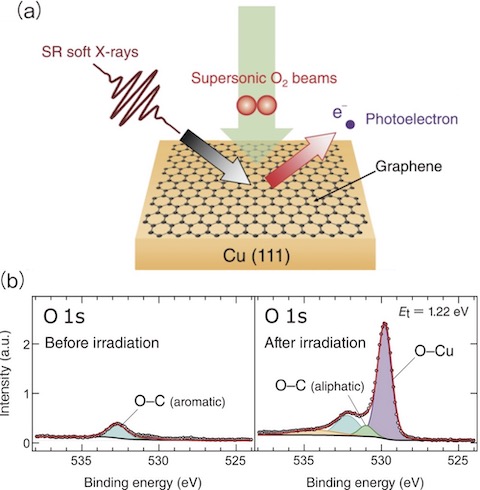
Fig.5-12 Gas barrier experiment by using real-time synchrotron radiation X-ray photoelectron spectroscopy (SR-XPS) combined with supersonic molecular beams and experimental evidence of O2 permeation through graphene
Fig.5-13 An image of an oxygen molecule passing through graphene (a net made of a single layer of carbon atoms)
Graphene, a carbon-atom net, has been attracting attention as a next-generation functional material because of its excellent physical properties such as low electrical resistance. Due to its unique physical properties, the Nobel Prize in Physics was awarded in 2010 for its discovery. Furthermore, as it is known that graphene has excellent gas barrier property, it is expected to be used as a protective film for easily oxidized transition metal catalysts (Ni, Cu, etc.). However, details of gas barrier properties remain unclear.
As is well known, the majority of O2 in an environment such as the atmosphere have kinetic energy (Et) of about 26 meV, however there are also small amounts of O2 with higher energies, which may overcome activation barriers in reaction pathways. Real-time SR-XPS using X-rays at 700 eV was applied to analyze the graphene/Cu(111) after O2 irradiation with various Et controlled by the supersonic molecular beams technique (Fig.5-12(a)). All experiments were conducted at the surface experimental station of the JAEA soft X-ray beamline (BL23SU) at SPring-8.
Fig.5-12(b) shows O 1s photoelectron spectra before (left) and after (right) irradiation for O2 with 1.22 eV. An increase in the peak exhibiting surface oxidation was clearly observed. A similar change in the spectrum was observed for O2 higher than 0.8 eV. Furthermore, it was found that the gas barrier property was maintained even after permeation. In addition to synchrotron experiments, computer simulations clarify that the permeation of O2 with higher energies occurs nondestructively with the aid of defects in graphene (Fig.5-13). The discovery of nondestructive transmission of O2 with high incident energies is expected to be applied to molecular filters. Since high-speed O2 are all around us, it is expected to produce a new type of protective films that prevent food deterioration, metal rusting and so on.
This research is part of the results of a joint research project with Tohoku University entitled “Synchrotron Radiation Realtime Photoemission Spectroscopic Study of Material Surface Processes”.
(Akitaka Yoshigoe)