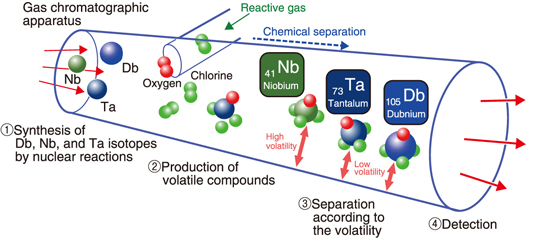
Fig.3-2 Schematic diagram of the experiment
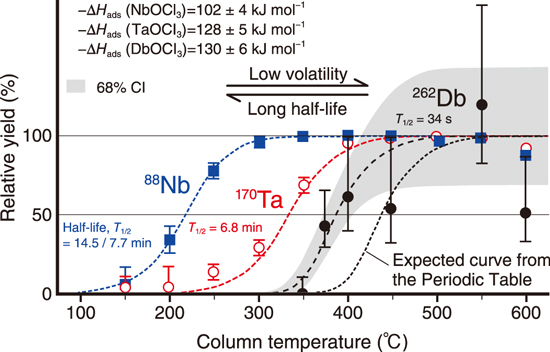
Fig.3-3 Changes of relative yields with the column temperature for the oxychlorides of Nb, Ta, and Db
Heavy elements (atomic number Z > 103) are called superheavy elements (SHEs). SHEs do not exist naturally on the Earth and are artificially synthesized in nuclear reactions. In such heavy elements, the velocity of the inner electrons moving around the nucleus approaches the speed of light, thereby eventually affecting the outermost electron orbits that characterize their chemical properties (relativistic effects). It has been pointed out that the chemical properties of the SHEs may be different from those expected according to the Periodic Table.
Dubnium (Db, Z = 105) is an SHE that belongs to Group 5 elements and is a homolog of niobium (Nb) and tantalum (Ta) in the Periodic Table. The production rate of Db is extremely low (about 1 atom per 5 min), and its half-life is quite short (34 s for 262Db). Hence, only one atom can be used at a time. The chemical properties of Db have not been investigated sufficiently due to the experimental difficulties.
In this work, we synthesized a Db isotope by using the JAEA tandem accelerator and performed a chemical analysis of its volatile compound using an on-line gas chromatographic apparatus. The volatility of the Db compound was compared with the values for the Nb and Ta compounds (Fig.3-2).
We successfully produced and separated dubnium oxychloride (DbOCl3) and clarified that its volatility was comparable to that of the Ta compound, though it was expected to be lower than that of the Ta compound from the simple extrapolation of the Periodic Table (Fig.3-3). This unexpected volatility of Db is explained by considering the strong relativistic effect.
The results of this research were featured on the back cover of the “Angewandte Chemie”. This achievement is expected not only to lead to an understanding of the Periodic Table, but also to contribute to the improvement of the accuracy of theoretical chemical calculations, which are increasingly being used in molecular design and material science in recent years.
(Tetsuya K. Sato)