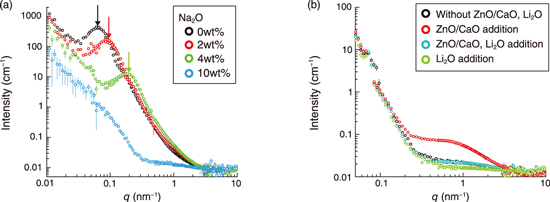
Fig.5-11 Small-angle neutron scattering (SANS) data of borosilicate glasses specialized for nuclear waste vitrification
We investigated the nanoscopic structure of borosilicate glasses to store for high-level radioactive liquid waste (HLLW) in the presence of typical additives Na2O and CaO/ZnO with and without Li2O by using the small-angle neutron scattering (SANS) technique. These additives have been practically used to lower the glass melting point, suppress macroscopic phase separation, and increase the chemical durability of glasses. Although the previous works successfully performed the structural analyses, they focus on length scales smaller than 1 nm. Hence, further study is necessary to identify the characteristics and microscopic structures of the glasses on the nano- and mesoscales even without HLLW.
Fig.5-11(a) shows the SANS profiles of the borosilicate glasses containing Na2O solutions of various concentrations. The scattering maximum (indicated by thick arrows) originates from the microphase separation between SiO2- and B2O3-rich domains on the nanoscale. The fraction of SiO2 in the borosilicate glass was much larger than that of B2O3, and thus, the B2O3 domains were likely to be dispersed in the SiO2 matrix. The peak position, qm, reflects the average distance between B2O3-rich domains, D, because D ≈ 2p/qm. The peak shifts toward a higher q value with increasing Na2O fraction, indicating an increase in D from 90 nm (0wt%; black) to 28 nm (4wt%; green). The peak completely disappeared at 10wt% (blue), indicating a miscible binary mixture. It is expected that each nanodomain tends to incorporate a specific component of HLLW into the glass framework.
Fig.5-11(b) shows the SANS profiles of samples. Using these profiles, the effect of CaO/ZnO addition with and without Li2O on the nanoscopic structure of the borosilicate glasses was examined. The SANS profile of the borosilicate glass containing CaO/ZnO additives (red) shows a shoulder peak, indicating the appearance of precipitates of approximately 2 nm size in the glass. Interestingly, CaO/ZnO addition was expected to suppress macroscopic phase separation; contrary to this belief, our result suggested that it induced inhomogeneities on the nanoscale. The shoulder peak was not present in the blue SANS profiles corresponding to CaO/ZnO addition with Li2O, or in the green profile corresponding to Li2O alone. Thus, the coaddition of Li2O with CaO/ZnO reduced the nanoscopic inhomogeneity of the glasses. This result suggests that CaO/ZnO should only be used in homogeneous glasses in conjunction with Li2O. We believe that the structural findings reported herein will contribute to the development of vitrification techniques for HLLW disposal treatment.
This work was supported by JSPS KAKENHI Grant-in-Aid for Scientific Research (B) (JP18H01921).
(Ryuhei Motokawa)