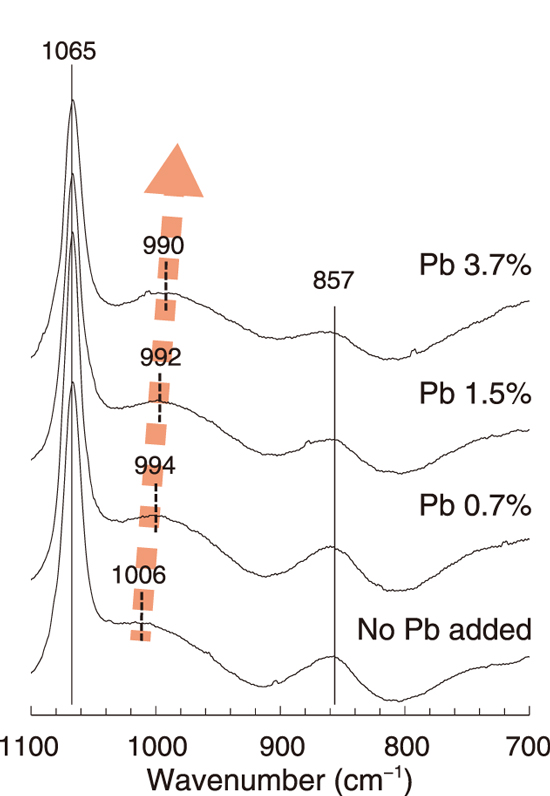
Fig.8-9 Shifts of silica–oxygen bonding in the aluminosilicate structure
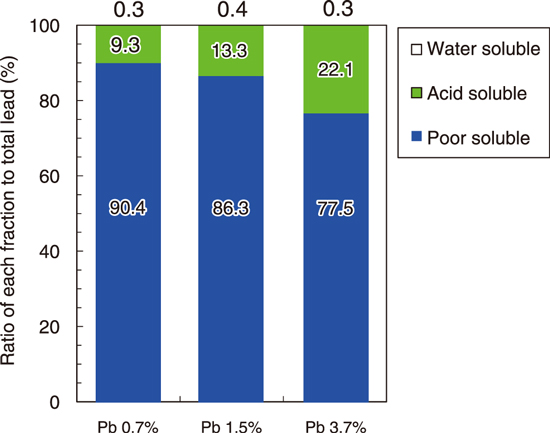
Fig.8-10 Leachability of lead solidified in alkali-activated materials (AAMs)
Lead is widely used as a radiation-shielding material in nuclear facilities. However, it is designated as a hazardous substance in industrial wastes, and there are regulations on the amount of leachate from wastes. Although there are no regulations on the hazardous substances contained in radioactive wastes yet, the development of technology to stabilize hazardous and radioactive wastes is required to reduce the environmental impact of disposal.
The JAEA has been conducting research and development on stabilization/solidification of hazardous substances into waste forms to dispose hazardous and radioactive wastes safely. Alkali-activated materials (AAMs) are amorphous inorganic materials consisting of silica and aluminum, and they harden via a mechanism different from cementation. In addition, it was reported that AAMs can stabilize cations into a matrix. This work investigated the characterization of lead stabilized in AAMs and the applicability of AAMs as a solidification material for hazardous and radioactive wastes. The results indicated that lead is stabilized as a part of the amorphous structure of AAMs.
AAMs were prepared by using chemical reagents, and lead chloride was added with lead ratios of 0.7, 1.5, and 3.7 mass% in solidified samples. Fig.8-9 shows that in the Raman spectra of solidified samples, the chemical bonding of silica to oxygen shifts depending on the lead concentration. Furthermore, the results of X-ray diffraction analysis and thermogravimetric analysis showed that lead was stabilized in an aluminosilicate matrix of the AAMs. Fig.8-10 shows that more than 75 mass% of lead in the solidified samples existed in a poorly soluble fraction in the leaching test with gradually changing strength of the acid in the solution. Lead was leached to less than 1% in a water-soluble fraction, and the remaining lead was extracted in an acid-soluble fraction. Thus, lead was stabilized as a part of an amorphous structure and existed in a poorly soluble form. These results indicate the possibility of developing a solidification/stabilization technique with the good confinement of hazardous substances.
Based on the results, the AAMs were evaluated for their applicability as a stabilization/solidification material, and the conditioning technology for hazardous and radioactive wastes has also been developed.
This work was conducted jointly with the Kyoto University under the title “Research on treatment of hazardous and radioactive wastes and changes in waste forms”.
(Junya Sato)