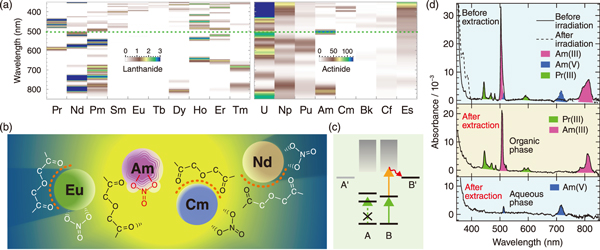
Fig.1 Concept of the laser-assisted separation and a typical experimental result
Spent nuclear fuel contains various lanthanide and actinide elements, also called f-block elements. Among them, the selective recovery of highly toxic ones, e.g., americium, is expected to lead to a significant reduction in the waste volume and management period in a geological repository. Meanwhile, the selective recovery of valuable elements such as neodymium, may boost the domestic supply of rare earth by facilitating its recycling. However, it is extremely difficult to separate f-block elements from each other by chemical processes because of their similar ion sizes. The primary difference among them is seen in the electronic configuration of the inner shell, which causes a prominent difference in the absorption spectra in the visible region with a number of sharp peaks at various wavelength (Fig.1(a)). If we can change chemical state of a specific element that is specified or “locked on” by laser light of wavelength tuned to the absorption peak of the element (Fig.1(b)), the locked-on element can be selectively recovered. However, the chemical state of these elements cannot be changed easily by the lock-on operation alone because the photon energy in the visible region is insufficient for such chemical reactions.
To promote the chemical reaction following a lock-on operation, we explored a two-step photoexcitation scheme-resonance-enhanced multiphoton excitation (Fig.1(c)). In actual experiments, a nitrous acid solution containing trivalent americium, Am(III), and praseodymium, Pr(III), was irradiated by intense laser light of wavelength 503 nm, which corresponds to the most intense absorption peak of Am(III). The resulting oxidation of Am(III) to the pentavalent state, Am(V), was observed, while no reaction was observed for Pr(III). Subsequently, after solvent extraction, only Am(V) was left in the aqueous phase and the other ions were transferred to the organic phase (Fig.1(d)), thus demonstrating the selective recovery of Am(III) via the laser lock-on operation.
In this study, the resonance-enhanced multiphoton excitation of actinides was observed for the first time and was applied for f-block element separation. The promising high selectivity between neighboring elements, e.g., between americium and curium, may reduce the burden of radioactive waste management and facilitate the maturation of a recycling-oriented society.
This study was conducted in collaboration with Osaka Metropolitan University and Institute for Laser Technology and supported by JSPS KAKENHI Grant-in-Aid for Early-Career Scientists (JP20K19999).
(Keiichi Yokoyama)