Fig.3-32 Comparison of detritiation system concept using catalyst and bacteria
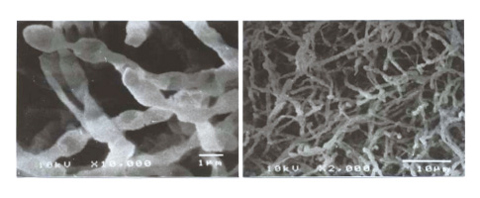
Fig.3-33 Typical tritium oxidizing bacteria (by SEM)

Fig.3-34 Bio-reactor for detritiation
A fusion reactor uses deuterium and tritium (3H) as a fuel. 3H is a radioisotope, emits a weak β-ray, and decays to 3He with a half-life of 12.3 years. It can be handled safely in a multiple confinement system, because it has characteristics similar to hydrogen. In fusion facilities like "ITER", the confinement functioning is always checked. Also, even if some 3H leaks into the buildings, that 3H is oxidized by a high temperature catalytic reactor and the tritiated water vapor is recovered by a dryer, in order to avoid 3H release to the environment as much as possible. This conventional detritiation system has been used extensively, but it requires keeping an expensive catalyst bed at high temperature (Fig.3-32). Therefore, R&D has been continuing at JAEA to develop a more efficient system. In this study, we took notice of research at Ibaraki Univ. (Prof. Ichimasa et al.), in which the almost all the hydrogen in an environment was oxidized by ordinary bacteria in the soil at room temperature. Recently, for the first time in the world, we succeeded in efficient atmosphere detritiation using bacteria which can be an alternative detritiation technology.
Traditionally, bacteria have been used extensively for fermentation in food, and medical products industries, but here we investigated the original idea that the special function of bacteria can be used for fast oxidation of hydrogen in atmosphere. We succeeded in finding and incubating bacteria with high 3H oxidation ability from the forest soil (Fig.3-33), and identifying the conditions making this bacteria active. Using this bacteria, a bio-reactor was fabricated (Fig.3-34), and tested it by connecting it to the Caisson Assembly for Tritium safety Study (CATS). We developed a bio-reactor that could oxidize atmospheric 3H in the same way as the catalytic reactor, with a detritiation factor of more than 85%. The ITER requirement of 99% should be attainable using multiple bio-reactor connection series. Further, the bacteria maintained about 70% of their 3H oxidation activity even after one year storage under 4°C. From the above accomplishments, we see a good possibility that the bio-reactor can be used for an alternative detritiation system. If this technology is used, a high temperature catalytic reactor would not be required, and the fabrication & operation cost and waste volume would be decreased. For the future practical use of bio-reactors, we will investigate their long-term performance, and also attempt using bacteria for processing of gas other than tritium.