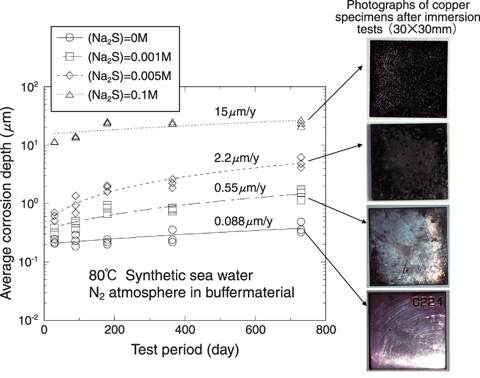
Fig.2-8 Influence of sulfide concentration on the corrosion of pure copper
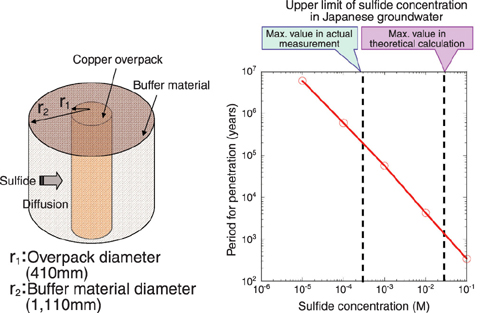
Fig.2-9 Schematic of the model for the evaluation of corrosion depth and the corrosion lifetime estimated by the model as a function of sulfide concentration
Overpack is a container for high-level radioactive waste disposal which is expected to prevent the contact of groundwater with vitrified waste at least for 1000 years. We have studied possible overpack materials, carbon steel in particular, and basic knowledge for designing the overpack including corrosion behavior has been obtained. However, the reliability of the repository system will be improved if a lifetime of overpack far beyond 1000 years could be achieved. Carbon steel is sufficiently strong to ensure a lifetime of 1000 years, since the corrosion rate in deep underground environment has been estimated to less than several µm/y. However, it is difficult to achieve a much longer lifetime, over tens of thousands of years, with carbon steel. Therefore, we have investigated alternative metals such as titanium and copper as candidate materials for the longer lifetime overpack.
Among the candidate overpack materials, copper is an only metal that is immune to corrosion under anaerobic conditions. The deep underground environment has inherently low oxygen concentration so that copper have a potential to achieve semi-permanent non-corrosion subject to change in the environmental conditions. However, such chemical stability of copper is known to be lost in the presence of sulfide (H2S, HS- and S2-). Therefore, we have been studying the effect of sulfide concentration on the corrosion behavior of copper under anaerobic conditions. As an example of our experimental results, the relationship between the sulfide concentration and the corrosion rate is shown in Fig.2-8. It was confirmed that the corrosion rate of copper increased with sulfide concentration. In case of 0.001M sulfide, the corrosion rate was less than 1µm/y, and linear extrapolating the experimental data yields and estimate of less than 1 mm corrosion in 1000 years. Since the copper overpack will be designed to have corrosion allowance of several centimeters, longer lifetime far beyond 1000 years could be achieved. In order to make the long term evaluation for an actual repository system, we performed a model calculation of long term corrosion depth assuming the present repository design. The buffer material which consists primarily of bentonite was installed around the overpack, so that the sulfide would contact the overpack surface only by diffusion through the buffer material. Consequently, the corrosion rate will be controlled by diffusion rate of sulfide in buffer material. The sulfide flux to the overpack surface was converted into the corrosion rate and the overpack lifetime was estimated as a function of sulfide concentration. As shown in Fig.2-9, if sulfide concentration is not over 0.0003M, the upper limit of actual measured data for general groundwater in Japan, a lifetime of several hundred thousand years is possible. However, if some kind of peculiar effect such as microbial action is assumed, the sulfide concentration can theoretically rise to 100 times higher. In such case, a very long lifetime cannot be expected.
Although copper have a potential of achieving an extremely long lifetime depending on the environmental conditions as above, it is necessary to clarify observe the environmental conditions and its long-term evolution if copper is selected as an overpack material. We are planning to extend our knowledge of the corrosion of copper in order to contribute to material selection, overpack designing and assessment of its long term integrity.