Fig.14-17 Outline of systems for 99Mo production by solution irradiation method, and evaluation items
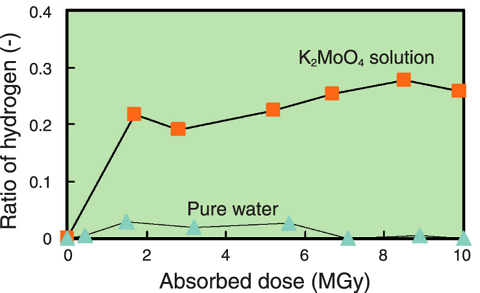
Fig.14-18 Relationship between absorbed dose of K2MoO4 solution and ratio of hydrogen in generated gas
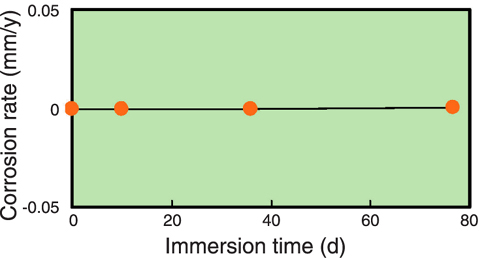
Fig.14-19 Relationship between immersion time and corrosion rate of SS304
Technetium-99m (99mTc) is the most widely used radiopharmaceutical for medical diagnosis. The raw material of 99mTc is molybdenum-99 (99Mo), and all 99Mo used in Japan is imported from foreign countries. However, a problem has emerged that the supply of 99Mo is unstable due to aging production facilities. Therefore, the stable supply and production of 99Mo is needed in Japan.
99Mo has been produced mainly by the fission method using highly enriched uranium. In Japan, 99Mo production by the fission method is difficult because of nuclear nonproliferation requirements and radioactive waste. Therefore, taking a hint from the shipment form of 99Mo, the solution irradiation method was invented (Fig.14-17). In this method, a solution of molybdate that has high solubility is irradiated with neutrons in a nuclear reactor. 99Mo is thus produced by the 98Mo (n,γ) 99Mo reaction and is collected with the high-performance Mo adsorbent PZC. This method has three advantages: 99Mo production rate can be increased simply by increasing the volume of the irradiation target, the processes required to produce a shipment are simpler, and the amount of radioactive waste is much smaller than that of the fission method.
As the molybdate solution used in the solution irradiation method, potassium molybdate (K2MoO4) solution which has a high concentration of molybdenum was selected to increase the 99Mo production rate. For the investigation of its suitability as the irradiation target, γ-ray irradiation tests of the solution were carried out. The test results indicated: (1) the ratio of hydrogen in the gas generated from the solution is 20 times higher than that from pure water (Fig.14-18); however, this is not a serious problem for the gas disposal device installed into the 99Mo production system, (2) the increase in calorific value of the solution upon γ-ray irradiation is about the same as that of pure water, (3) the compatibility (nonreactivity) between the solution and stainless steel material of the capsule is very good (Fig.14-19), (4) the solution flows stably in a pipe 4mm in inner diameter and 30m in length without precipitation.
From the above results, it is clear that the K2MoO4 solution is suitable as the irradiation target for the 99Mo production system by the solution irradiation method. In addition, it is evaluated that over 20% of the domestic demand for 99Mo (88.8TBq/week) can be provided by this system using JMTR. Based on this obtained data, plans for the practical application of this system are to be considered.