
Fig.7-9 Appearance of corrosion testing apparatus
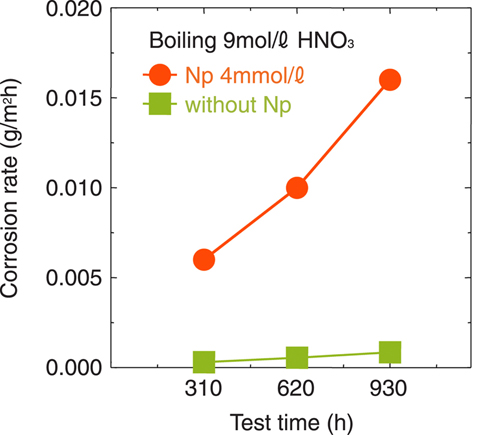
Fig.7-10 Corrosion rate of stainless steel in boiling HNO3 solution at 70°C
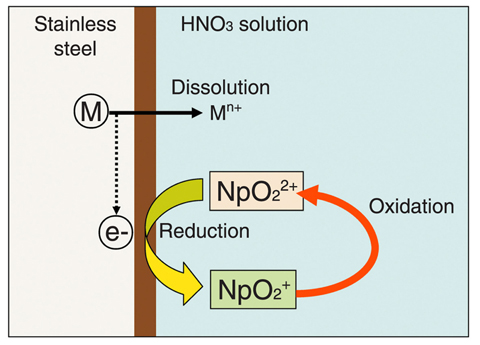
Fig.7-11?ESchematic illustration of corrosion mechanism in HNO3 solution with neptunium
Usable nuclear fuels (uranium and plutonium) are left in spent nuclear fuels from a nuclear power plant. A plant which chemically collects usable nuclear fuel from spent fuel, separates fission products, and safely treats radioactive wastes is called a spent fuel reprocessing plant. In Japan, there are two such plants, in Tokai-mura and Rokkasho-mura. Uranium is a limited fuel like oil. The recycling of nuclear fuel is very important for sustainable production and utilization of nuclear energy.
A very corrosive boiling nitric acid solution is used to dissolve nuclear fuel in a reprocessing plant. The solution contains plutonium and ruthenium which are dissolved from spent nuclear fuel. It has been known that these elements strongly corrode commercial grade stainless steel, and special corrosion-resistant stainless steels are used in reprocessing plants. Recently, it has been reported that a small amount of neptunium (Np) also promotes the corrosion of the stainless steel. The mechanism of corrosion acceleration by Np was investigated. The tests were conducted in the Waste Safety Testing Facility (WASTEF) that could handle radioactive materials such as spent nuclear fuel. Fig.7-9 shows the appearance of the corrosion testing apparatus set in a hot cell in WASTEF.
Fig.7-10 shows the corrosion rate of stainless steel which was immersed in boiling nitric acid (HNO3) solution at 70°C under reduced pressure. The solution contained a small amount of Np (ca.4mmol/L), and the concentration of HNO3 was 9mol/L. The corrosion rate of stainless steel in the HNO3 solution with Np was about 10 times higher than that without Np. This corrosion acceleration by Np was investigated using electrochemical and spectrochemical analyses that could examine the influence of ionic species in solutions. As shown in Fig.7-11, Np6+ (NpO22+) caused the dissolution of a metal in the stainless steel and itself was thereby reduced to Np5+ (NpO2+). The Np5+ was re-oxidized to Np6+ in the HNO3 solution. It was found that this cycle led to the increase in the corrosion rate of stainless steel in HNO3 solutions with Np. We have been developing the new stainless steels, which are very corrosion-resistant to boiling HNO3 solutions with corrosive ionic species such as Np.
The above knowledge is being utilized as basic data for the safe operation of Rokkasho reprocessing plant.