
Fig.7-12 Module for TRU high temperature chemistry
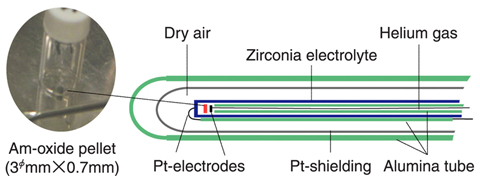
Fig.7-13 Electrochemical cell with a zirconia electrolyte
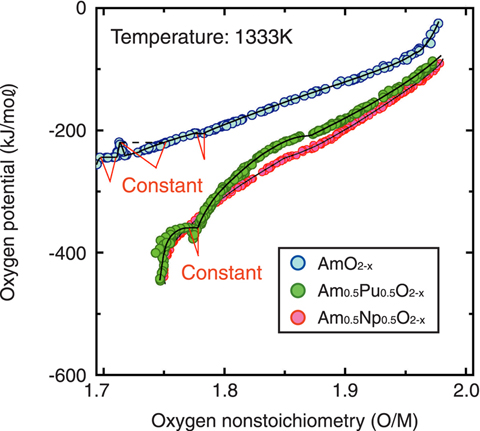
Fig.7-14 The relation between the oxygen potentials and oxygen nonstoichiometry (O/M) of Am oxides
For future nuclear fuel cycles, we are developing the technology of recovering minor actinides (MA: Np, Am and Cm) from spent fuels and transmuting them by fast reactors. MA recycling can reduce the long-term hazard and the area of the site needed for disposal of the high-level radioactive waste. To burn MA effectively and safely, it is necessary to develop the MA-containing oxide fuels and get a reasonable understanding of their oxygen potentials to predict their compatibility with fuel-cladding and their thermal behavior during irradiation.
Therefore, we installed a module for TRU high temperature chemistry (Fig.7-12) in NUCEF of JAEA, in order to study MA-containing fuels. We investigated the relation between the oxygen potentials and oxygen nonstoichiometry (O/M) of Am oxides by an electrochemical method (Fig.7-13). In this study, we could test a sufficiently large amount of Am, and electrochemically control O/M of the samples with sufficient accuracy to get data with good precision (Fig.7-14). We found that the thermodynamic properties of Am oxides exhibit a systematic change. This study indicates a method for prediction of the thermal behavior of Am-containing oxide fuels, enabling the evaluation of technological feasibility of MA recycling.
This study includes the results of the collaborative research program "TRU (transuranium elements) Behavior in Oxide Fuels" with Tohoku Electric Power Company, Tokyo Electric Power Company and the Japan Atomic Power Company, and the results of "Development of Common and Fundamental Technologies for Evaluation of Nuclear Fuel Behavior in MA recycling" entrusted to the Japan Atomic Energy Agency by the Ministry of Education, Culture, Sports, Science and Technology of Japan (MEXT).