
Fig.6-11 Image of a silica membrane
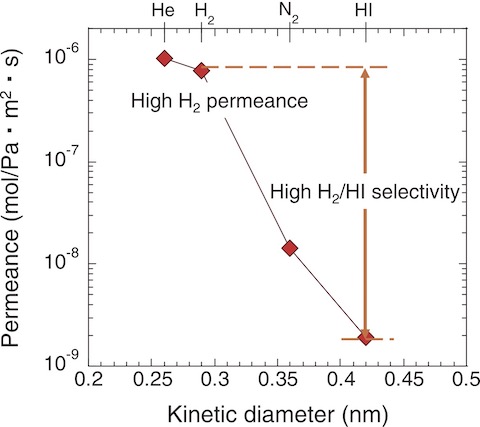
Fig.6-12 Single-gas permeation performance of the silica membrane
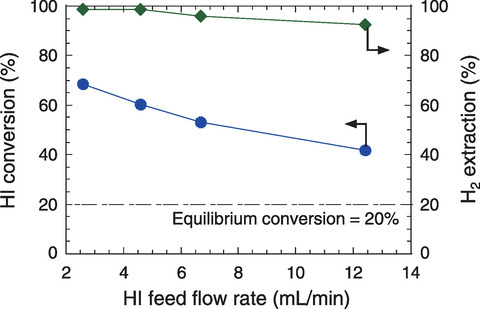
Fig.6-13 HI decomposition performances of the silica membrane reactor at T = 400 ℃ and HI feed flow rates of 2.6-12.4 mL/min
The thermochemical sulfur-iodine (IS) process is studied by the JAEA to realize massive hydrogen production using nuclear heat at high temperature. This process consists of three coupled chemical reactions:
Bunsen reaction (ca. 100 ℃):
| SO2 + I2 + 2H2O → H2SO4 + 2HI | (1) |
Sulfuric acid decomposition (ca. 850 ℃):
| H2SO4 → H2O + SO2 + 0.5O2 | (2) |
Hydrogen iodide decomposition (ca. 400 ℃):
| 2HI → H2 + I2 | (3) |
The HI decomposition (Eq. 3) is a key reaction in the IS process. However, the equilibrium conversion of HI to H2 and I2 is very low (e.g., approximately 20% at 400 ℃), leading to an increase in the amount of recycled materials and a decrease in the thermal efficiency of the process. The usage of a membrane reactor that combines a catalytic reaction and H2 separation in one unit can shift the equilibrium and enhance the HI conversion. The membrane reactor demonstration comprises two stages: i) fabrication of a membrane possessing high H2 separation performance and high corrosion stability; and ii) reactor assembly with membrane and catalyst integration.
In this study, silica membranes consisting of a three-layer structure comprising a porous α-alumina ceramic support, an intermediate γ-alumina layer, and a top silica layer that is H2 selective were prepared via the counter-diffusion chemical vapor deposition of hexyltrimethoxysilane (Fig.6-11).
Fig.6-12 presents the He, H2, N2, and HI permeances through the developed silica membranes. The silica membranes exhibited high H2 permeance and high H2/HI selectivity and were stable in the corrosive gas of HI for 11 h. Therefore, the research extended to the assembly of a membrane reactor with the integration of the silica membrane and active carbon catalyst and investigated its potential for H2 production from the HI decomposition.
Fig.6-13 shows the results of the HI decomposition tests varying the HI feed flow rate. The influence of the HI feed flow rate on the membrane reactor performance was significant in the lower range of the flow rate. Moreover, better than 70% conversion of the HI decomposition and 98% H2 extraction were attained. The results suggest that the developed membrane reactor equipped with the silica membrane would reduce the recycled flow rate in the HI decomposition by up to approximately 80% and improve the total thermal efficiency of the IS process by 1%.
We are currently working on the improvement of the thermal efficiency of the IS process by developing a membrane reactor equipped with industrial-size silica membranes in HI decomposition.
(Odtsetseg Myagmarjav)