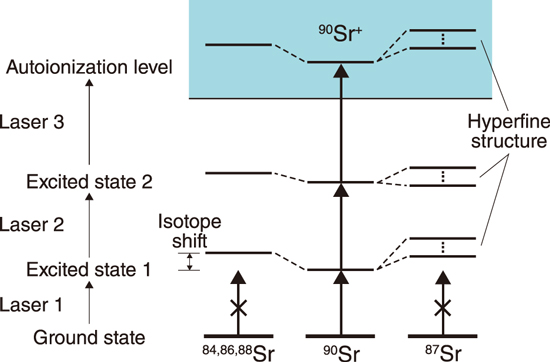
Fig.1-27 Element and isotope-selective laser resonance ionization of 90Sr atoms
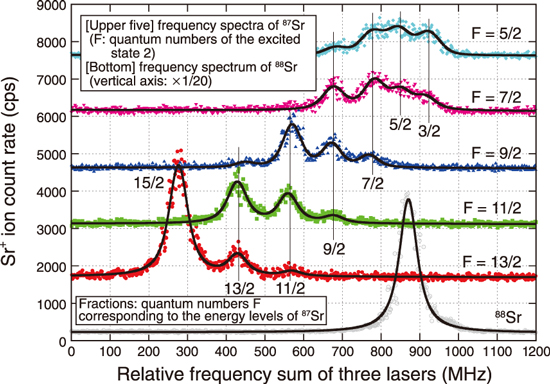
Fig.1-28 Example of the observed 88Sr and 87Sr frequency spectra
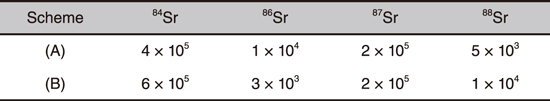
Table 1-2 Measured optical isotopic selectivities of 90Sr with respect to stable Sr isotopes (84Sr, 86Sr, 87Sr, and 88Sr)
Strontium 90 (90Sr, half-life of 28.8 years) is one of the major radionuclides that was released into the environment during the TEPCO’s Fukushima Daiichi NPS accident in 2011. Because its biochemical behavior is similar to that of calcium, 90Sr tends to accumulate in the human bones, causing long-term internal exposure.
Several methods have been reported so far for determining 90Sr. These methods are classified into radiation measurement and mass spectrometry. The former is the conventional method of detecting β radiation from 90Y, and it requires a time-consuming process of 90Y separation; therefore, it is considered unsuitable for rapid analysis. The latter is a method of detecting 90Sr+ ions after ionization, and an example is inductively coupled plasma mass spectrometry (ICP-MS). However, ICP-MS suffers from isobaric interference caused by zirconium 90 (90Zr), and mass spectral interference of 88Sr in samples with high concentrations of stable Sr isotopes.
In this work, we developed a device for measuring Sr+ ions using a quadrupole mass spectrometer, where three wavelength-tuned lasers are used for element- and isotope-selective laser resonance ionization of 90Sr atoms, as shown in Fig.1-27. Isotope shifts of stable Sr isotopes (84Sr, 86Sr, 87Sr, and 88Sr) and energy-level splitting of 87Sr due to the hyperfine structure were observed for each of the two ionization schemes: (A) 689.4 nm–487.4 nm–393.8 nm and (B) 689.4 nm–472.4 nm–404.2 nm, both of which are expected to have high isotopic selectivity. Fig.1-28 shows an example of the observed 88Sr and 87Sr frequency spectra using scheme (B) where Laser-3 (404.2 nm) frequency was scanned. The measured 90Sr optical isotopic selectivities with respect to the stable Sr isotopes ranged from 103 to 105 for both schemes, as shown in Table 1-2. Compared to the existing mass spectrometry, our method is expected to greatly simplify the sample pretreatment process, especially in the analysis of soil and marine samples containing large amounts of 90Zr and stable Sr isotopes, respectively.
A part of this work was supported by the JAEA Nuclear Energy S&T and Human Resource Development Project through concentrating wisdom (JPJA18B18071760), “Development of technology for rapid analysis of strontium-90 with low isotopic abundance using laser resonance ionization”.
(Yoshihiro Iwata)